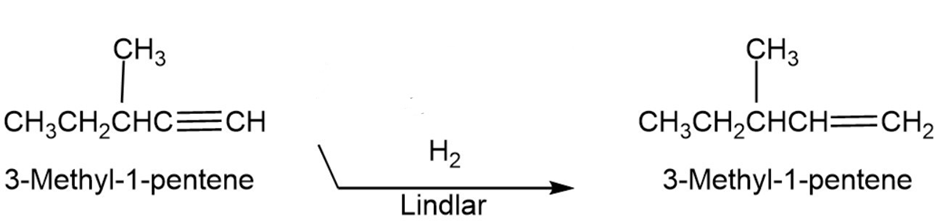
Chapter 9: Q. 9-9-8 P-c (page 274)
Using any alkyne needed, how would you prepare the following alkenes?
(c) 3-Methyl-1-pentene
Short Answer
Learning Materials
Features
Discover
Chapter 9: Q. 9-9-8 P-c (page 274)
Using any alkyne needed, how would you prepare the following alkenes?
(c) 3-Methyl-1-pentene

All the tools & learning materials you need for study success - in one app.
Get started for free
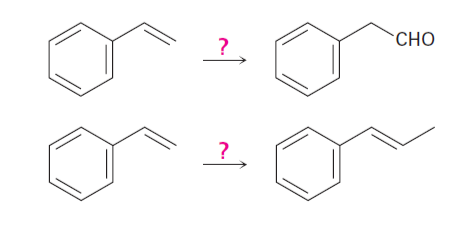
How would you carry out the following conversions? More than one
step is needed in each case.
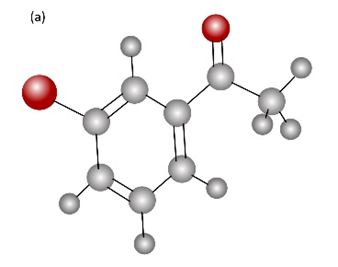
How would you prepare the following carbonyl compounds starting from an alkyne (reddish brown Br)?
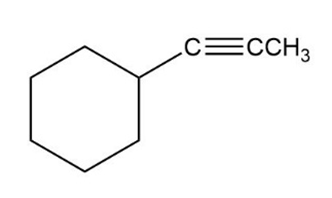
Show the terminal alkyne and alkyl halide from which the following products can be obtained. If two routes look feasible, list both.
What products would you expect from the following reactions?
a.
Terminal alkynes react with and water to yield bromo ketones. For example:
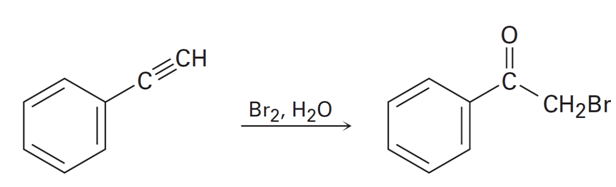
Propose a mechanism for the reaction. To what reaction of alkenes is
the process analogous?
What do you think about this solution?
We value your feedback to improve our textbook solutions.