Chapter 9: Q9-9P a (page 276)
The of acetone, of acetone, is 19.3. Which of the following bases is strong enough to deprotonate acetone?
(a)KOH (pkaof H2O 15.7)
Short Answer
Answer
Not strong enough to deprotonate acetone due to less than acetone.
Learning Materials
Features
Discover
Chapter 9: Q9-9P a (page 276)
The of acetone, of acetone, is 19.3. Which of the following bases is strong enough to deprotonate acetone?
(a)KOH (pkaof H2O 15.7)
Answer
Not strong enough to deprotonate acetone due to less than acetone.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: How would you carry out the following conversions? More than one
step may be needed in some instances.
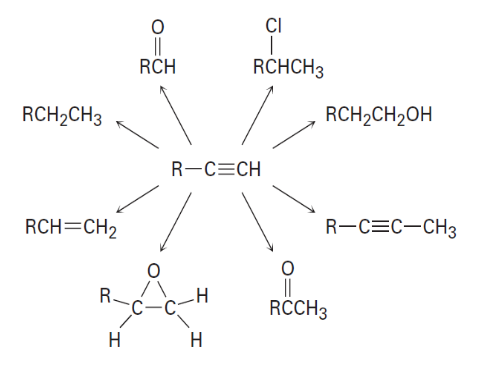
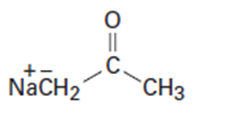
Which of the following bases could be used to deprotonate 1-butyne?
d)
Synthesize the following compounds using 1-butyne as the only source of carbon along with any inorganic reagents you need. More than one step may be needed.(a) 1,1,2,2- Tetrachlorobutane(b) 1,1- Dichloro-2-ethylcyclopropane
What products would you expect from the following reactions?
c.
The pkaof acetone, CH3COCH3, is 19.3. Which of the following bases is strong enough to deprotonate acetone?
(b)
What do you think about this solution?
We value your feedback to improve our textbook solutions.