Chapter 7: Q7-13P-c (page 197)
Assign E or Z configuration to the following alkenes:
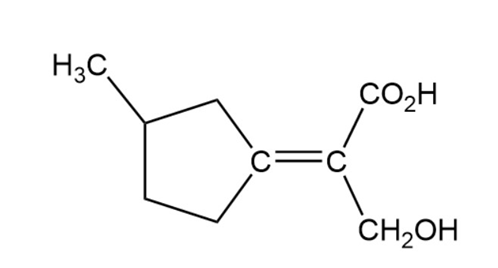
Short Answer
Z configuration
Learning Materials
Features
Discover
Chapter 7: Q7-13P-c (page 197)
Assign E or Z configuration to the following alkenes:

Z configuration
All the tools & learning materials you need for study success - in one app.
Get started for free
Name the following alkenes, and tell which compound in each pair is more stable:
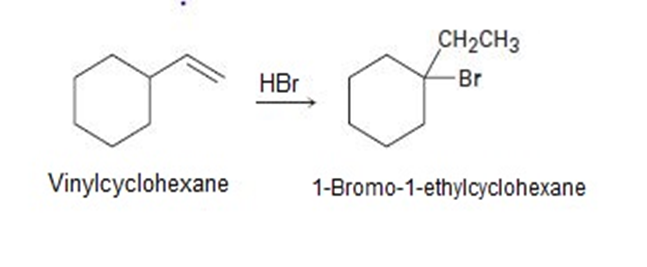
On treatment with HBr, vinylcyclohexane undergoes addition and rearrangement to yield 1-bromo-1-ethylcyclohexane. Using curved arrows, propose a mechanism to account for this result.
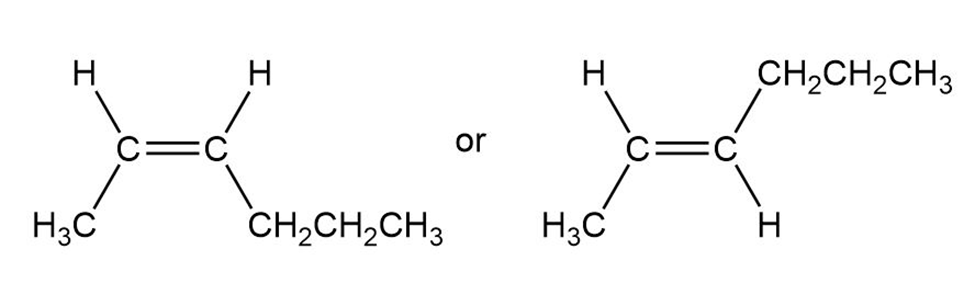
Which of the following compounds can exist as pairs of cis-trans isomers? Draw each cis-trans pair, and indicate the geometry of each isomer.
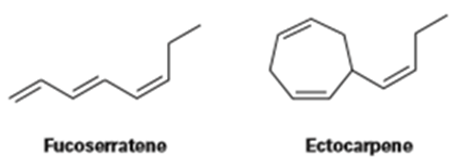
Fucoserratene and ectocarpene are sex pheromones produced by marine brown algae. What are their systematic names? (Ectocarpene is a bit difficult; make your best guess, and then check your answer in the Study Guide and Solutions Manual.)
Draw an energy diagram for the addition of HBr to 1-pentene. Let one curve on your diagram show the formation of 1-bromopentane product and another curve on the same diagram show the formation of 2-bromopentane product. Label the positions for all reactants, intermediates, and products. Which curve has the higher-energy carbocation intermediate? Which curve has the higher-energy first transition state?
What do you think about this solution?
We value your feedback to improve our textbook solutions.