Chapter 7: Q7-21P (page 219)
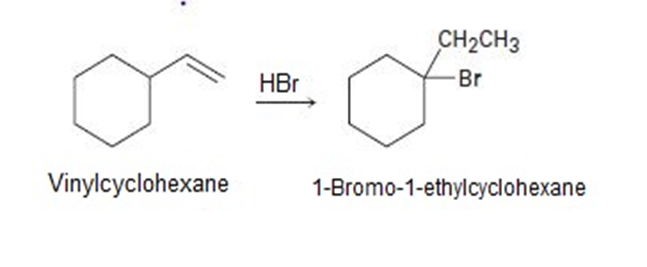
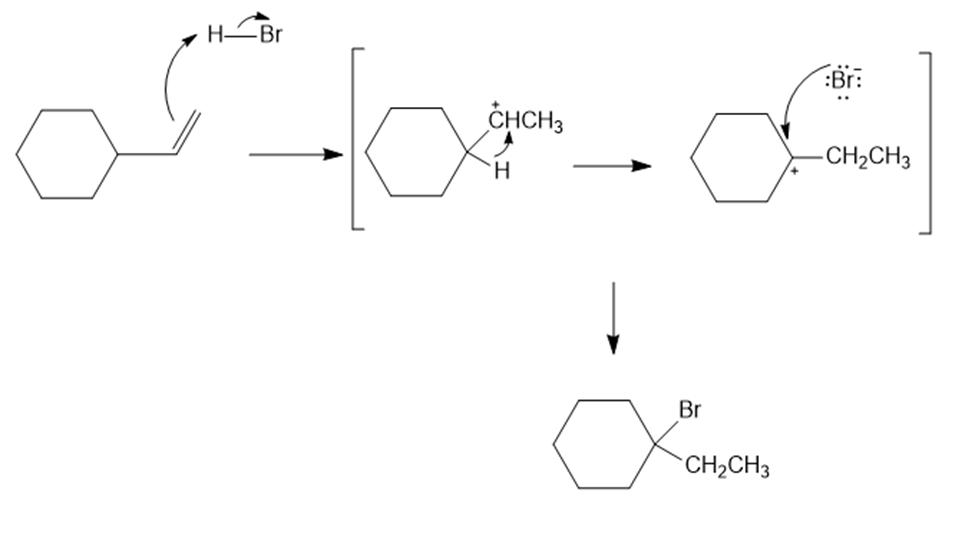
On treatment with HBr, vinylcyclohexane undergoes addition and rearrangement to yield 1-bromo-1-ethylcyclohexane. Using curved arrows, propose a mechanism to account for this result.
Short Answer
Learning Materials
Features
Discover
Chapter 7: Q7-21P (page 219)
On treatment with HBr, vinylcyclohexane undergoes addition and rearrangement to yield 1-bromo-1-ethylcyclohexane. Using curved arrows, propose a mechanism to account for this result.


All the tools & learning materials you need for study success - in one app.
Get started for free
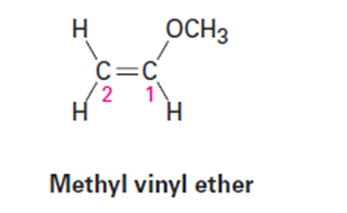
Question: When methyl vinyl ether reacts with a strong acid, the proton adds to
C2 exclusively, instead of C1 or the oxygen atom. Draw the three protonated
forms of methyl vinyl ether and explain this observation
Assign E or Z configuration to the following alkenes:
Sketch the transition-state structures involved in the reaction of HBr with 1-pentene (Problem 7-69). Tell whether each structure resembles reactant or product.
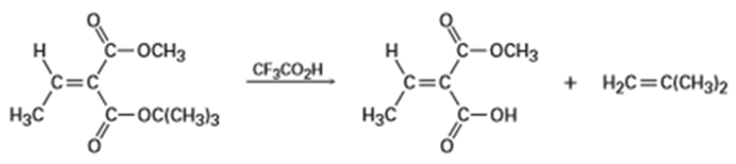
tert-Butyl [RCO2C(CH3)3] esters are converted into carboxylic acids [RC02H] by reaction with trifluoroacetic acid, a reaction useful in protein synthesis (Section 26-7). Assign E, Z designation to the double bonds of both reactant and product in the following scheme, and explain why there is an apparent change in double-bond stereochemistry:
Which of the following compounds can exist as pairs of cis-trans isomers? Draw each cis-trans pair, and indicate the geometry of each isomer.
What do you think about this solution?
We value your feedback to improve our textbook solutions.