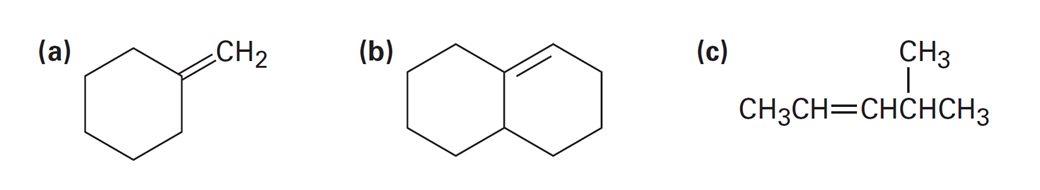
Chapter 7: Q7-13P-b (page 197)
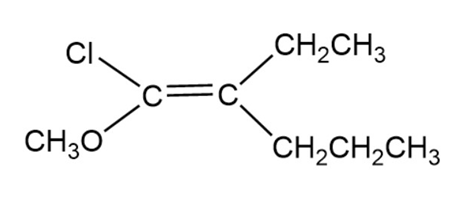
Assign E or Z configuration to the following alkenes:
Short Answer
E configuration
Learning Materials
Features
Discover
Chapter 7: Q7-13P-b (page 197)
Assign E or Z configuration to the following alkenes:

E configuration
All the tools & learning materials you need for study success - in one app.
Get started for free
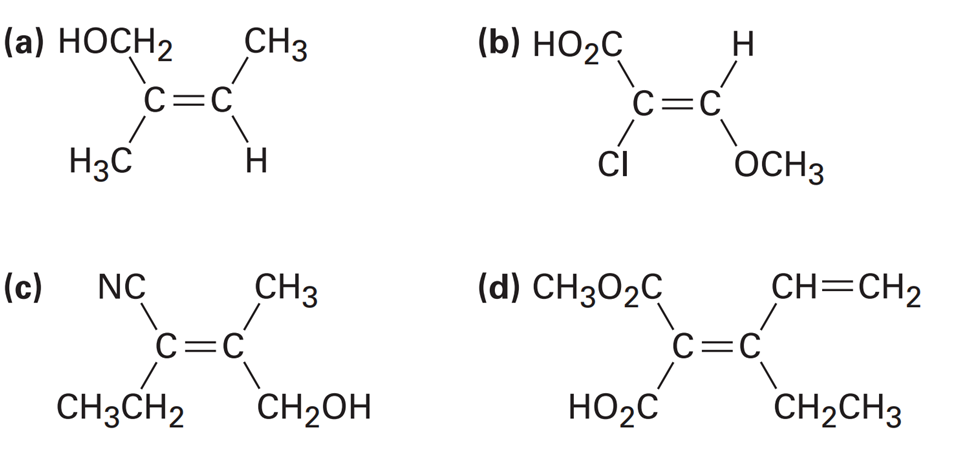
Question: Assign E or Z configuration to each of the following compounds:
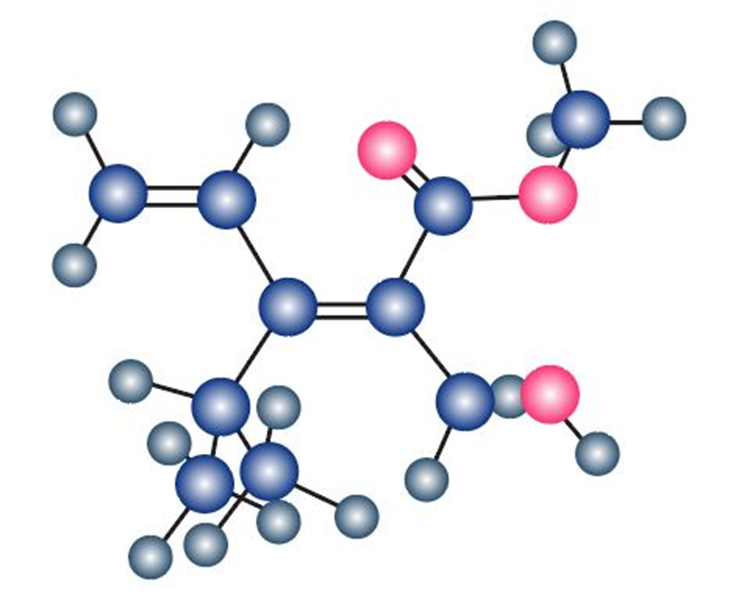
Assign stereochemistry (E or Z) to the double bond in the following compound, and convert the drawing into a skeletal structure (red = O):
Show the structures of the carbocation intermediates you would expect in the following reactions:
Reaction of 2,3-dimethyl-1-butene with HBr leads to an alkyl bromide,. On treatment of this alkyl bromide with KOH in methanol, elimination of HBr occurs and a hydrocarbon that is isomeric with the starting alkene is formed. What is the structure of this hydrocarbon, and how do you think it is formed from the alkyl bromide?
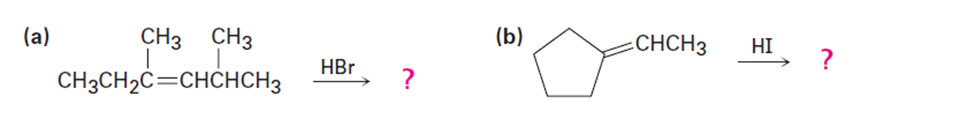
Predict the major product from addition of HBr to each of the following alkenes:
What do you think about this solution?
We value your feedback to improve our textbook solutions.