Chapter 7: Q51 (page 218)
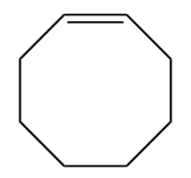
Q 51 Normally, a trans alkene is more stable than its cis isomer trans-Cyclooctene, however, is less stable than cis-cyclooctene by 38.5 kJ/mol. Explain.
Short Answer
Answer
Trans alkene is more stable than cis alkene due to the less steric strain present in the trans-isomers.