Chapter 1: Q57P (page 2)
Rank the bonds from the most polar to least polar.
a. C - O,C - F,C - N
b. C - Cl > C - Br > C - I
c. H - O,H - N,H - C
d. C - H,C - C,C - N
Short Answer
a. C-F>C-O>C-N
b. C-Cl>C-Br>C-I
c. H-O>H-N>H-C
d. C-N>C-H>C-C
Learning Materials
Features
Discover
Chapter 1: Q57P (page 2)
Rank the bonds from the most polar to least polar.
a. C - O,C - F,C - N
b. C - Cl > C - Br > C - I
c. H - O,H - N,H - C
d. C - H,C - C,C - N
a. C-F>C-O>C-N
b. C-Cl>C-Br>C-I
c. H-O>H-N>H-C
d. C-N>C-H>C-C
All the tools & learning materials you need for study success - in one app.
Get started for free
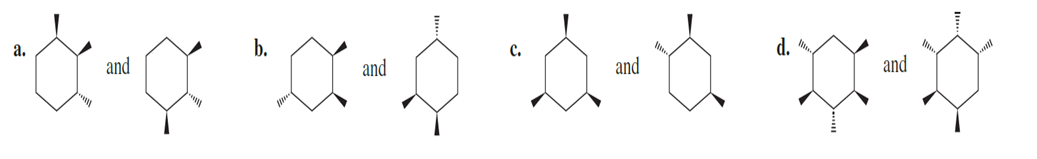
Question:Are the following pairs identical, enantiomers, diastereomers, or constitutional isomers?
Which of the following molecules would you expect to have a dipole moment of zero? To answer parts g and h, you may need to review the Problem Solving Strategy on p. 39.

According to the potential maps for ammonium ion, which atom has the greatest electron density?
Which of the following has
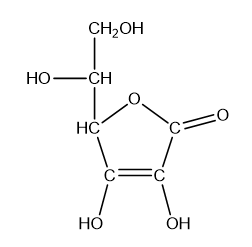
What is the hybridisation of each of the carbon and oxygen atoms in vitamin c?
What do you think about this solution?
We value your feedback to improve our textbook solutions.