Chapter 1: Q-1-34P (page 2)
According to the potential maps for ammonium ion, which atom has the greatest electron density?
Short Answer
The nitrogen atom in the ammonium ion has the greatest electron density.
Learning Materials
Features
Discover
Chapter 1: Q-1-34P (page 2)
According to the potential maps for ammonium ion, which atom has the greatest electron density?
The nitrogen atom in the ammonium ion has the greatest electron density.
All the tools & learning materials you need for study success - in one app.
Get started for free
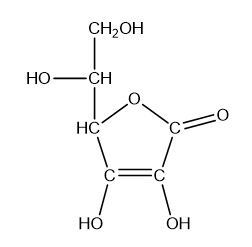
What is the hybridisation of each of the carbon and oxygen atoms in vitamin c?
Rank the carbanions shown in the margin from most basic to least basic.
 HC=C-
HC=C-
 H2C=CH-
H2C=CH-
 H3C-CH2-
H3C-CH2-
The compounds commonly known as “amino acids” are actually -aminocarboxylic acids (Section 21.0). What carbonyl compounds should be used to synthesize the two amino acids shown here?
Explain why CH3Cl has a greater dipole moment than CH3F even though F is more electronegative than Cl.
An unknown disaccharide gives a positive Tollens’ test. A glycosidase hydrolyzes it to d-galactose and d-mannose. When the disaccharide is treated with methyl iodide and Ag2O and then hydrolyzed with dilute HCl, the products are 2,3,4,6-tetra-O-methylgalactose and 2,3,4-tri-O-methylmannose. Propose
a structure for the disaccharide.
What do you think about this solution?
We value your feedback to improve our textbook solutions.