Chapter 23: The Organic Chemistry of the Coenzymes, Compounds Derived From Vitamins
Q10P
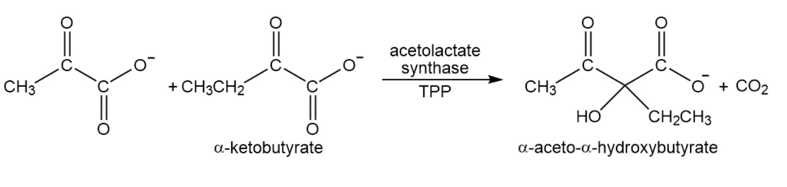
Acetolactatesynthasetransfers the acyl group of pyruvate to \(\alpha \)-ketobutyrate. This is the first step in the biosynthesis of the amino acid isoleucine. Propose a mechanism for this reaction.
Q12P
a. What acyl group does pyruvate decarboxylase transfer to a proton?
b. What acyl group does the pyruvate dehydrogenase complex transfer to coenzyme A?
c. What acyl group does transketolase transfer to ribose-5-P?
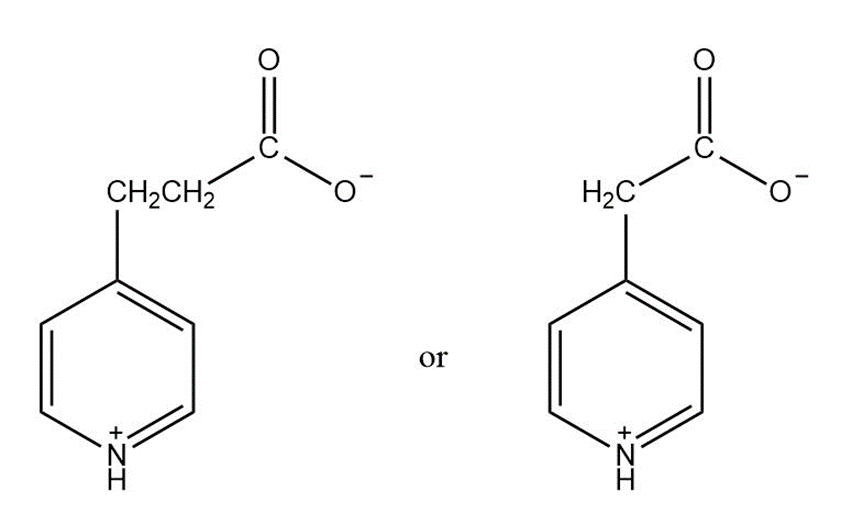
Q13 P
Which compound is more easily decarboxylated?
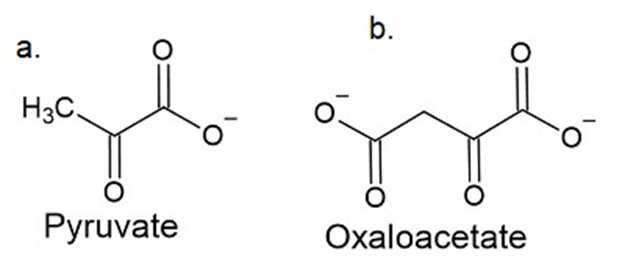
Q14 P
\(\alpha \)-Keto acids other than \(\alpha \) -ketoglutarate can accept the amino group from pyridoxamine in enzyme catalyzed transamination reactions. What amino acids are formed when the following a-keto acids accept the amino group?
Q15 P
Explain why the ability of PLP to catalyze an amino acid transformation is greatly reduced if a PLP- requiring enzymatic reaction is carried out at a pH at which the pyridine nitrogen is not protonated.
Q16 P
Explain why the ability of PLP to catalyze an amino acid transformation is greatly reduced if the OH substituent of pyridoxal phosphate is replaced by OCH3.
Q17 P
The enzyme that catalyzes the bond cleavage reaction that converts serine to glycine removes the substituent (R) bonded to the \(\alpha \)-carbon in the first step of the reaction. Starting with PLP bound to serine in an imine linkage, propose a mechanism for this reaction. (Hint:The first step involves removal of the proton from serine’s OH group.)
Q18 P
Propose a mechanism for the -elimination reaction shown on page 1082.
Q20P
Question:A fatty acid (a long straight-chain carboxylic acid with an even number of carbons) is metabolized to acetyl- CoA, which can then enter the citric acid cycle to be further metabolized (Section 24.9). A fatty acid with an odd number of carbons is metabolized to acetyl-CoA and one equivalent of propionyl-CoA. Propionyl-CoA cannot enter the citric acid cycle. Two coenzyme-requiring enzymes are needed to convert it to succinyl-CoA, a compound that can enter the citric acid cycle. Write the two enzyme-catalyzed reactions and include the names
of the required coenzymes.
Q21P
Question: How do the structures of tetrahydrofolate and aminopterin differ?
