Chapter 23: Q13 P (page 1085)
Which compound is more easily decarboxylated?
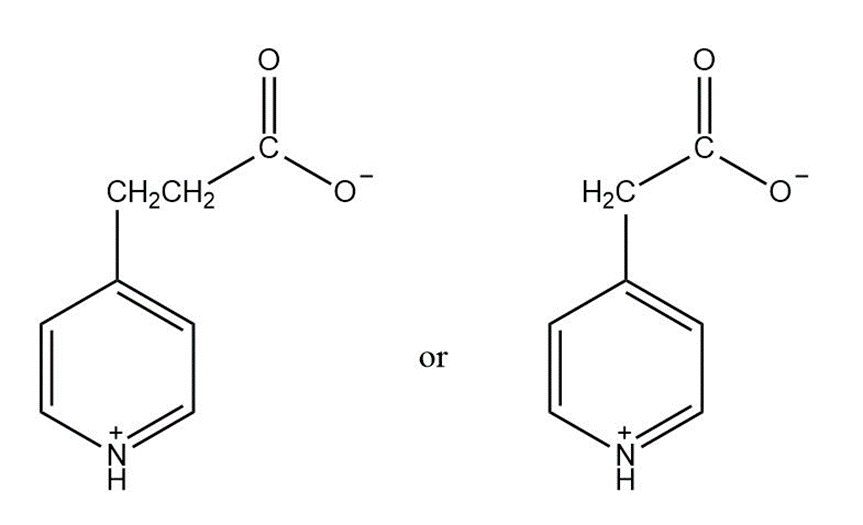
Short Answer
Compound 2 is much easier to decarboxylate than compound 1 because of resonance.
Learning Materials
Features
Discover
Chapter 23: Q13 P (page 1085)
Which compound is more easily decarboxylated?

Compound 2 is much easier to decarboxylate than compound 1 because of resonance.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: How do the structures of tetrahydrofolate and aminopterin differ?
How many conjugated double bonds are there in
a. FAD?
b. FADH2
Question:When UMP is dissolved in T2O, exchange of T for H occurs at the 5-position. Propose a mechanism for this exchange.
Draw structures that show the similarity between the decarboxylation of the pyruvate–TPP intermediate and the decarboxylation of a \(\beta \)-keto acid.
Explain why the ability of PLP to catalyze an amino acid transformation is greatly reduced if a PLP- requiring enzymatic reaction is carried out at a pH at which the pyridine nitrogen is not protonated.
What do you think about this solution?
We value your feedback to improve our textbook solutions.