Chapter 23: Q4p (page 1072)
How many conjugated double bonds are there in
a. FAD?
b. FADH2
Short Answer
Pyrrole is a much stronger acid than ammonia because of resonance.
Learning Materials
Features
Discover
Chapter 23: Q4p (page 1072)
How many conjugated double bonds are there in
a. FAD?
b. FADH2
Pyrrole is a much stronger acid than ammonia because of resonance.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: For each of the following reactions, name both the enzyme that catalyzes the reaction and the required coenzyme:
Question: What is the source of the methyl group in thymidine?
Question:A fatty acid (a long straight-chain carboxylic acid with an even number of carbons) is metabolized to acetyl- CoA, which can then enter the citric acid cycle to be further metabolized (Section 24.9). A fatty acid with an odd number of carbons is metabolized to acetyl-CoA and one equivalent of propionyl-CoA. Propionyl-CoA cannot enter the citric acid cycle. Two coenzyme-requiring enzymes are needed to convert it to succinyl-CoA, a compound that can enter the citric acid cycle. Write the two enzyme-catalyzed reactions and include the names
of the required coenzymes.
What are the products of the following reaction?
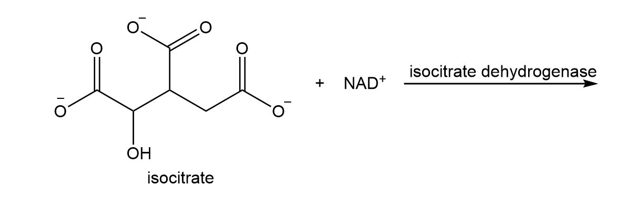
Draw structures that show the similarity between the decarboxylation of the pyruvate–TPP intermediate and the decarboxylation of a \(\beta \)-keto acid.
What do you think about this solution?
We value your feedback to improve our textbook solutions.