Chapter 23: Q2P (page 1069)
What are the products of the following reaction?
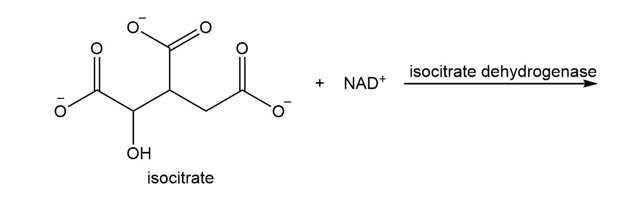
Short Answer
Learning Materials
Features
Discover
Chapter 23: Q2P (page 1069)
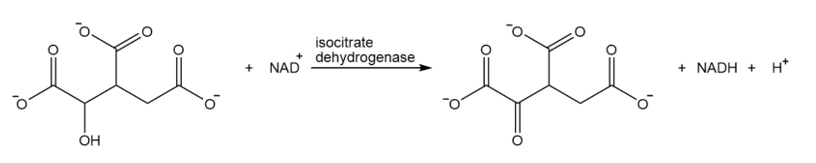
What are the products of the following reaction?


All the tools & learning materials you need for study success - in one app.
Get started for free
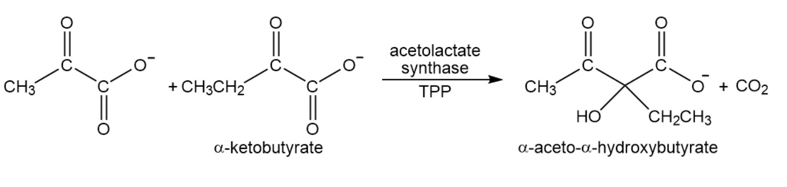
Acetolactatesynthasetransfers the acyl group of pyruvate to \(\alpha \)-ketobutyrate. This is the first step in the biosynthesis of the amino acid isoleucine. Propose a mechanism for this reaction.
Question:Five coenzymes are required by -ketoglutarate dehydrogenase, the enzyme in the citric acid cycle that converts -ketoglutarate to succinyl-CoA.
a. Identify the coenzymes.
b.Propose a mechanism for the reaction.
Explain why the hydrogens of the C-8 methyl group are more acidic than those of the C-7 methyl group.
Question:When UMP is dissolved in T2O, exchange of T for H occurs at the 5-position. Propose a mechanism for this exchange.
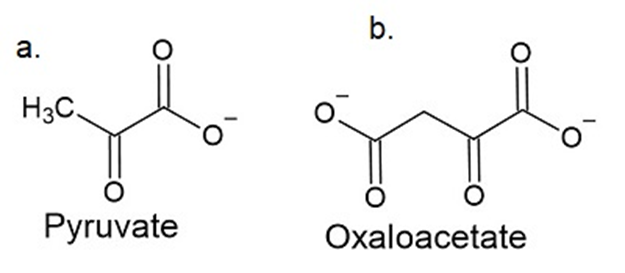
\(\alpha \)-Keto acids other than \(\alpha \) -ketoglutarate can accept the amino group from pyridoxamine in enzyme catalyzed transamination reactions. What amino acids are formed when the following a-keto acids accept the amino group?
What do you think about this solution?
We value your feedback to improve our textbook solutions.