Chapter 23: Q10P (page 1076)
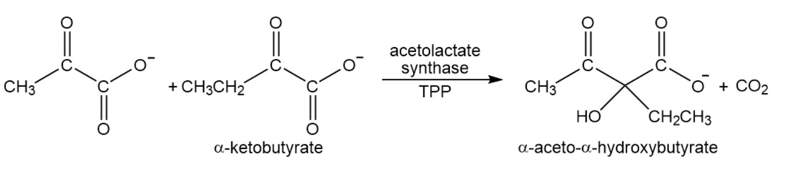
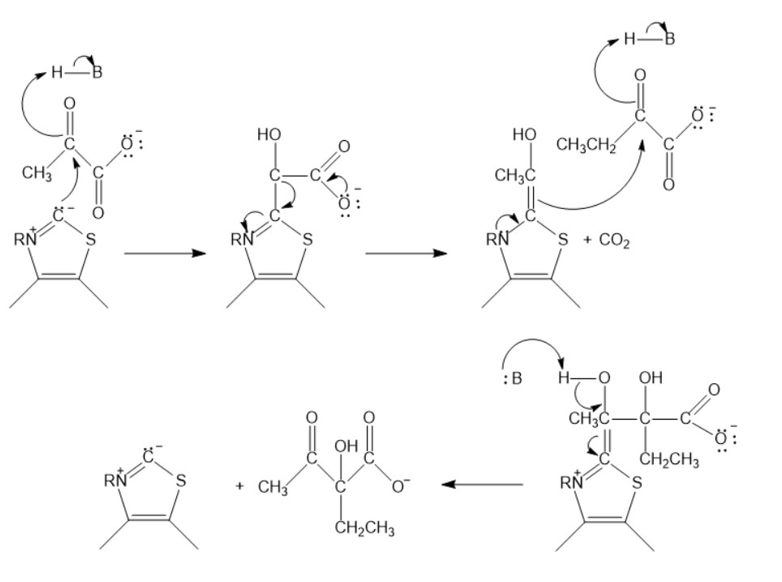
Acetolactatesynthasetransfers the acyl group of pyruvate to \(\alpha \)-ketobutyrate. This is the first step in the biosynthesis of the amino acid isoleucine. Propose a mechanism for this reaction.
Short Answer
Learning Materials
Features
Discover
Chapter 23: Q10P (page 1076)
Acetolactatesynthasetransfers the acyl group of pyruvate to \(\alpha \)-ketobutyrate. This is the first step in the biosynthesis of the amino acid isoleucine. Propose a mechanism for this reaction.


All the tools & learning materials you need for study success - in one app.
Get started for free
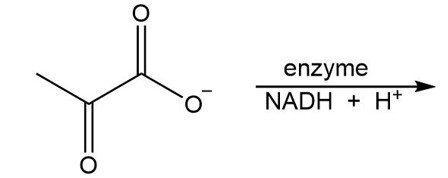
What is the product of the following reaction?
Instead of adding to the 4a position and protonating N-5, the thiolate ion could have added to the 10a position and protonated N-1. (The numbering system is on page 1071.) Why is addition to the 4a position favored? (Hint:Which nitrogen is a stronger base?)
Question:Draw the products of the following reaction, where T is tritium:
Question:Five coenzymes are required by -ketoglutarate dehydrogenase, the enzyme in the citric acid cycle that converts -ketoglutarate to succinyl-CoA.
a. Identify the coenzymes.
b.Propose a mechanism for the reaction.
Question: What is the source of the methyl group in thymidine?
What do you think about this solution?
We value your feedback to improve our textbook solutions.