Chapter 5: Alkenes: Structure, Nomenclature, and an Introduction to Reactivity, Thermodynamics and Kinetics
Q 10TP
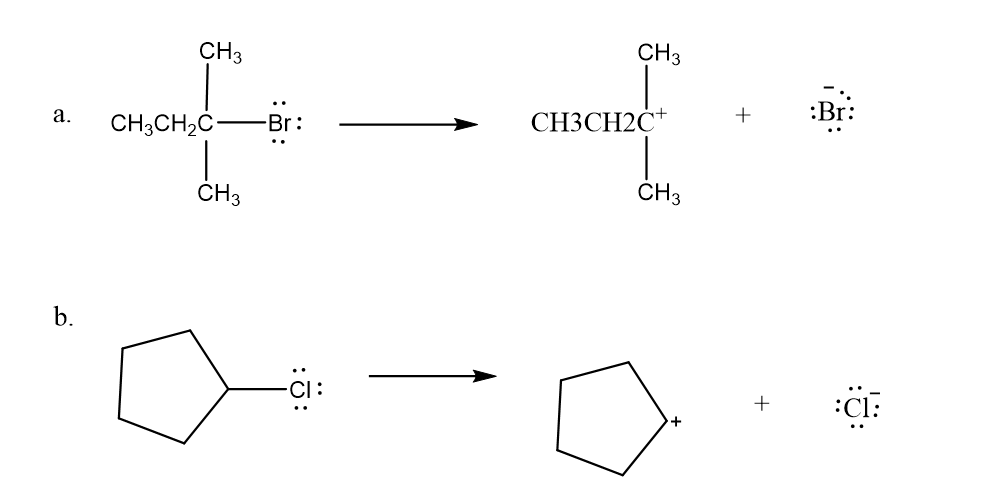
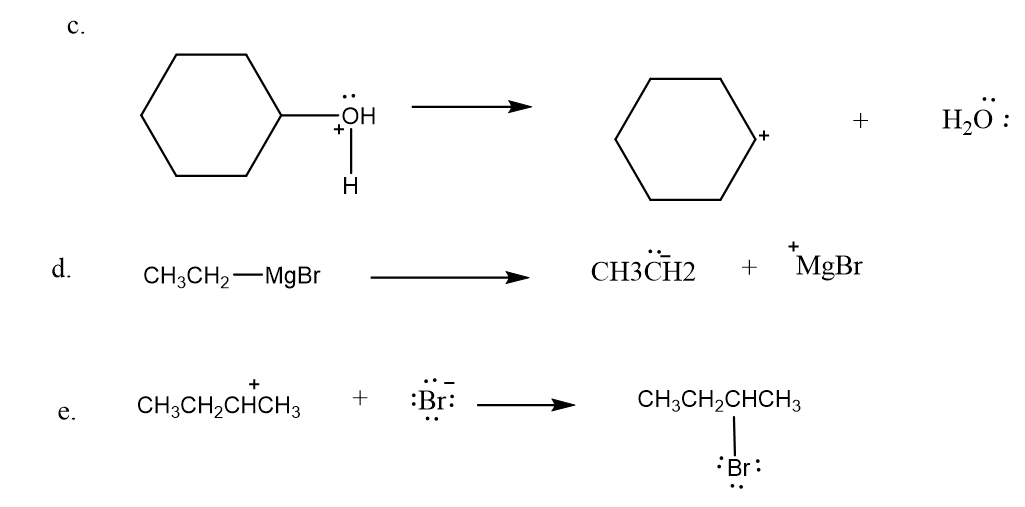
Use what the curved arrows tell you about electron movement to determine the product(s) of each reaction step.
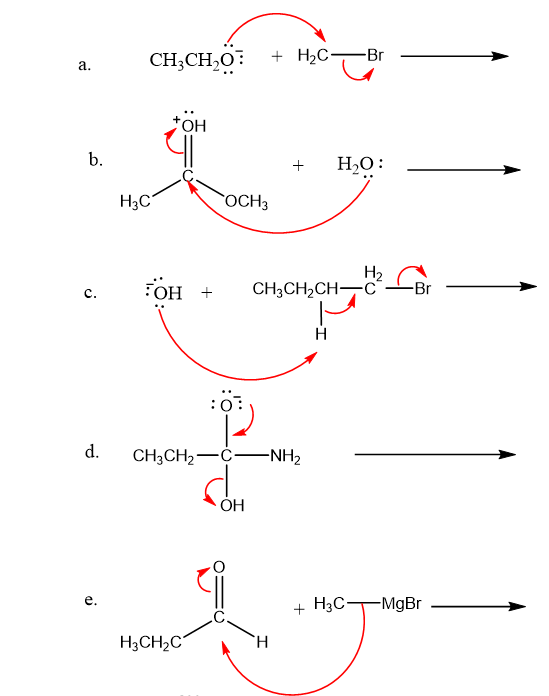
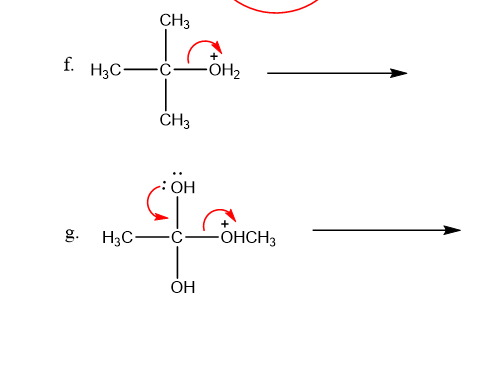
Q12P
Question: Draw the isomers for the following compounds and then name each one:
a. 2-methyl-2,4-hexadiene
b. 2,4-heptadiene
c. 1,3-pentadiene
Q13P
Question: Which of the following are electrophiles, and which are nucleophiles?
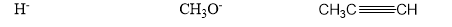

Q14P
Question: Identify the nucleophile and the electrophile in the following acid–base reactions:
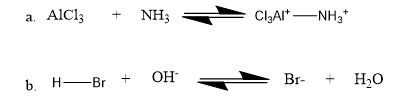
Q16 P
Draw the products that would be obtained by following the incorrect arrows in the box entitled “A Few Words about Curved Arrows” and explain what is wrong with the structures you obtain.
Q17P
Question: Draw the products that would be obtained by following the incorrect arrows in the box entitled “A Few Words about Curved Arrows” and explain what is wrong with the structures you obtain
Q18P
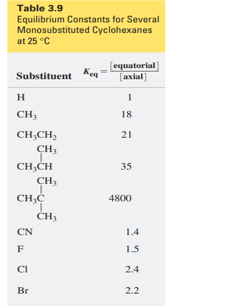
Question: a. Which of the monosubstituted cyclohexanes in Table 3.9 on p. 128 has a negative ∆G° for the conversion of an axial-substituted chair conformer to an equatorial-substituted chair conformer?
b.Which monosubstituted cyclohexane has the most negative ∆G° for this conversion?
c. Which monosubstituted cyclohexane has the greatest preference for an equatorial position?
d. Calculate ∆G° for the conversion of “axial” methylcyclohexane to “equatorial” methylcyclohexane at 25 °C.
Q1TP
Draw curved arrows to show the movement of the electrons in the following reaction steps.
Q20 P
a. Calculate the percentage of isopropylcyclohexane molecules that have the isopropyl substituent in an equatorial position at equilibrium. (Its ∆G° value at 25 °C is -2.1 kcal/mol.)
b. Why is the percentage of molecules with the substituent in an equatorial position greater for isopropylcyclohexane than for fluorocyclohexane?
Q21 P
a. For which reaction in each set will ∆S° be more significant?
b. For which reaction will ∆S° be positive?

