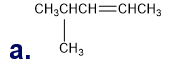
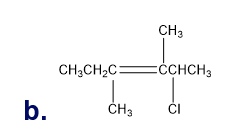
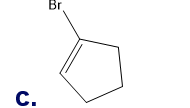
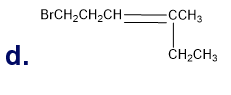
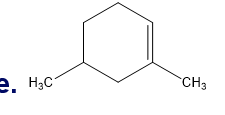
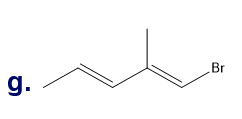
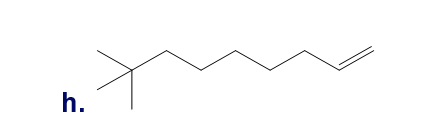
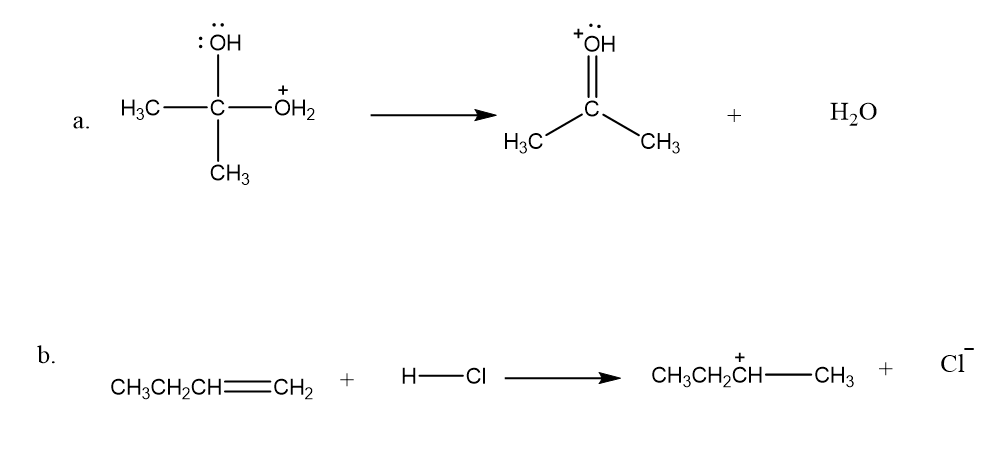
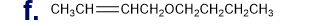Chapter 5: Q18P (page 204)
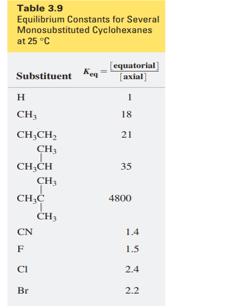
Question: a. Which of the monosubstituted cyclohexanes in Table 3.9 on p. 128 has a negative ∆G° for the conversion of an axial-substituted chair conformer to an equatorial-substituted chair conformer?
b.Which monosubstituted cyclohexane has the most negative ∆G° for this conversion?
c. Which monosubstituted cyclohexane has the greatest preference for an equatorial position?
d. Calculate ∆G° for the conversion of “axial” methylcyclohexane to “equatorial” methylcyclohexane at 25 °C.
Short Answer
- All of the equilibria have negative ∆G° values for the conversion of an axial-substituted chair conformer to an equatorial-substituted chair conformer.
- Tert-butylcyclohexane
- Tert-butylcyclohexane
- ∆G°=-1.7 kcal/mol