Chapter 5: Alkenes: Structure, Nomenclature, and an Introduction to Reactivity, Thermodynamics and Kinetics
Q5TP
Draw curved arrows to show the movement of the electrons that result in formation of the given product(s).
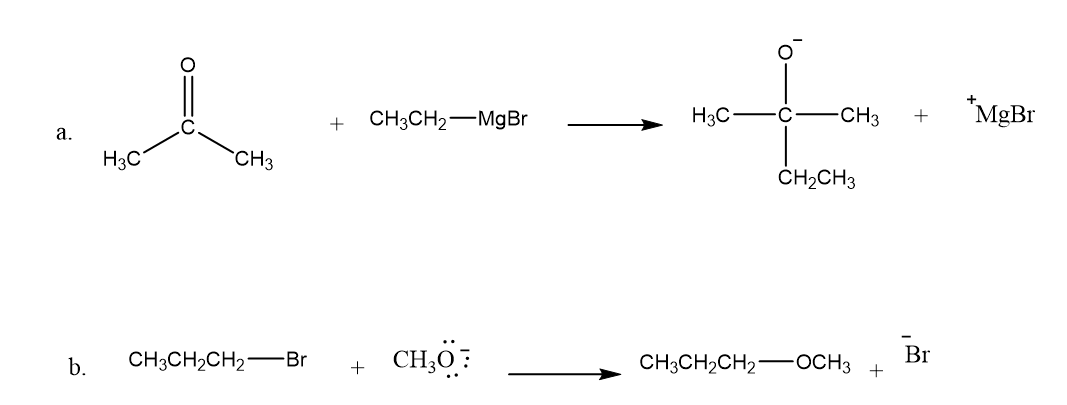

Q60P
Draw the structure of a compound with molecular C8H14 that reacts with one equivalent of H2 over Pd/C to form a meso compound.
Q61P
a. What is the equilibrium constant for a reaction that is carried out at 25°C (298 K) with ΔH° = 20 kcal/mol and ΔS° = 5.0 x 10-2 kcal mol-1 K-1?
b. What is the equilibrium constant for the same reaction carried out at 125°C?
Q62P
Using curved arrows, show the mechanism of the following reaction:

Q63P
For a reaction carried out at 25°C with an equilibrium constant of 1 × 10−3, in order to increase the equilibrium constant by a factor of 10:
a. How much must ΔG° change?
b. How much must ΔH° change if ΔS° = 0 kcal mol-1 K-1?
c. How much must ΔS° change if ΔH° = 0 kcal mol-1?
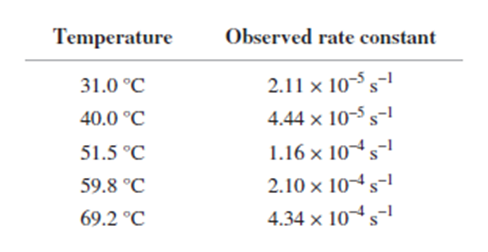
Q65P
From the following rate constants, determined at five temperatures, calculate the experimental energy of activation and ΔG±,Δ��±,���Ի�Δ��±for the reaction at 30°C:
Q6TP
Draw curved arrows to show the movement of the electrons that result in formation of the given product(s).
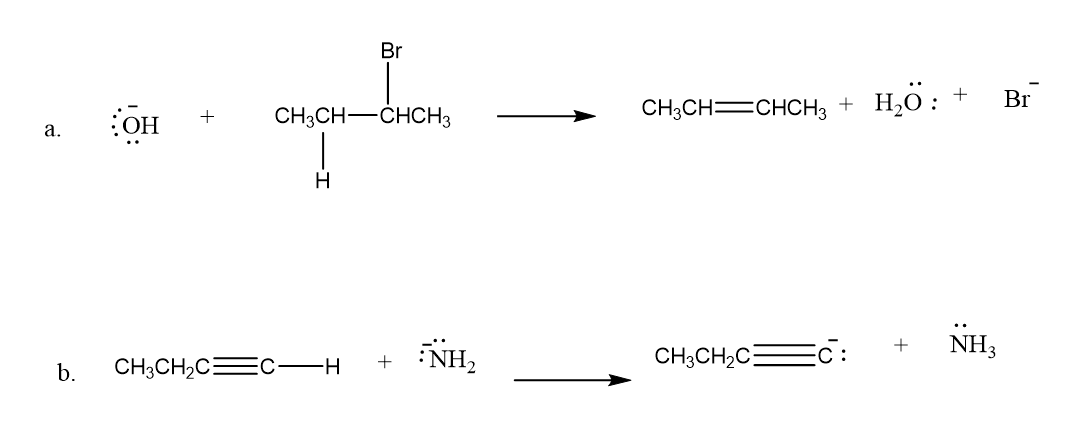

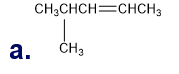
Q7P
What is each compound’s systematic name?
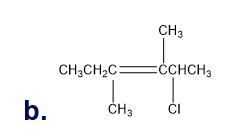
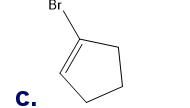
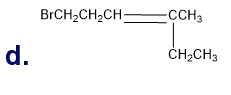
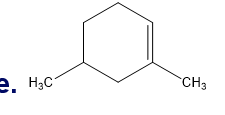
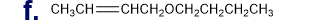
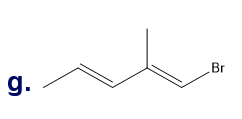

Q7TP
Draw curved arrows to show the movement of the electrons that result in formation of the given product(s).
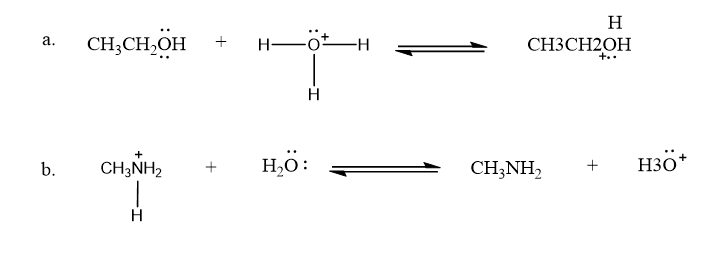
Q8P
Question: a. How many vinylic hydrogens does cyclopentene have?
b. How many allylic hydrogens does it have?
