Chapter 5: Q7TP (page 190)
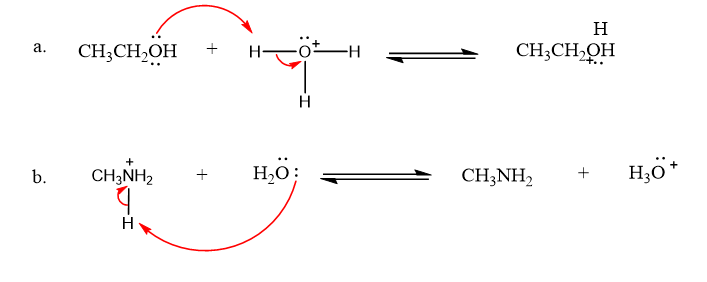
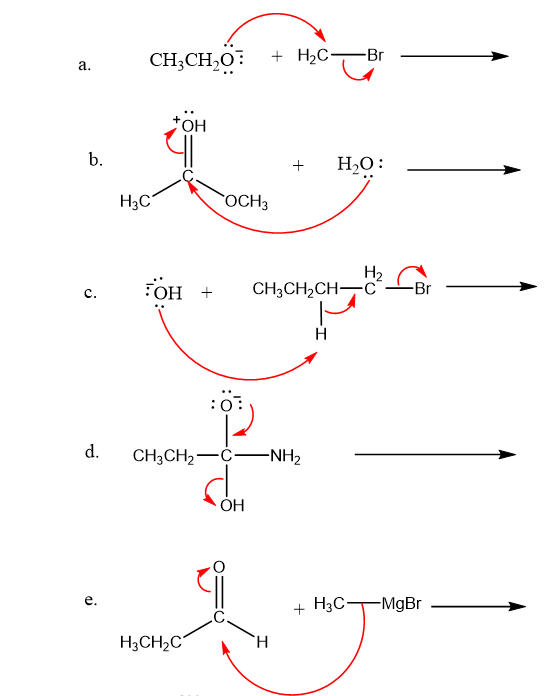
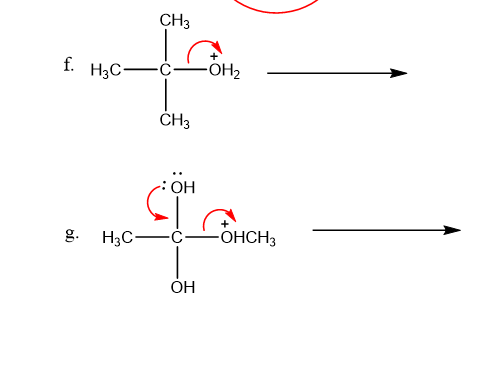
Draw curved arrows to show the movement of the electrons that result in formation of the given product(s).
Short Answer
Learning Materials
Features
Discover
Chapter 5: Q7TP (page 190)
Draw curved arrows to show the movement of the electrons that result in formation of the given product(s).


All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Draw the structure for each of the following:
a. 3,3-dimethylcyclopentene
b. 6-bromo-2,3-dimethyl-2-hexene
c. ethyl vinyl ether
d. allyl alcohol
Question: Draw the isomers for the following compounds and then name each one:
a. 2-methyl-2,4-hexadiene
b. 2,4-heptadiene
c. 1,3-pentadiene
a. Calculate the percentage of isopropylcyclohexane molecules that have the isopropyl substituent in an equatorial position at equilibrium. (Its ∆G° value at 25 °C is -2.1 kcal/mol.)
b. Why is the percentage of molecules with the substituent in an equatorial position greater for isopropylcyclohexane than for fluorocyclohexane?
Draw the structure of a hydrocarbon that has six carbon atoms and
a. three vinylic hydrogens and two allylic hydrogens.
b. three vinylic hydrogens and one allylic hydrogen.
c. three vinylic hydrogens and no allylic hydrogens.
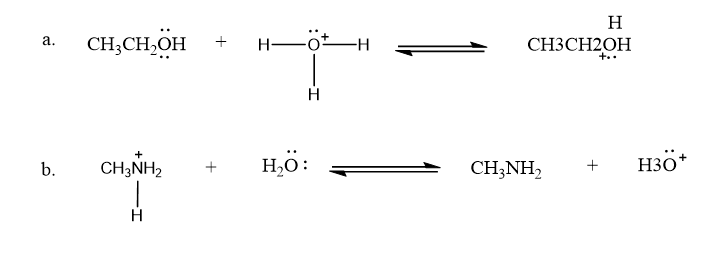
Use what the curved arrows tell you about electron movement to determine the product(s) of each reaction step.
What do you think about this solution?
We value your feedback to improve our textbook solutions.