Chapter 5: Q 10TP (page 190)
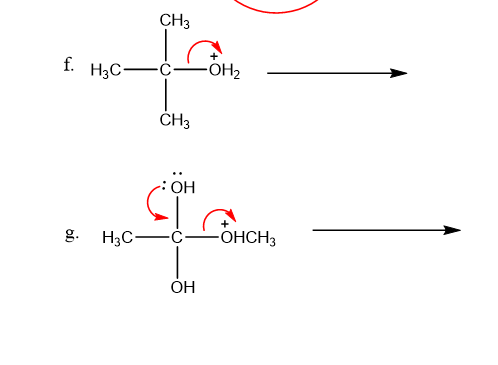
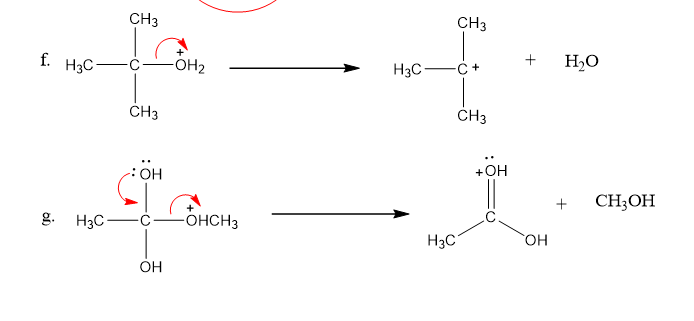
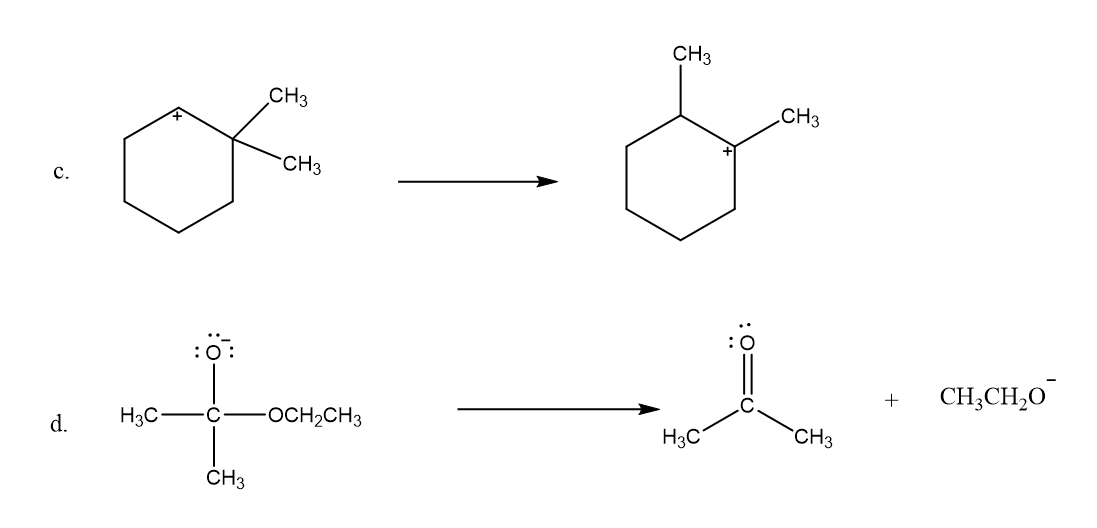
Use what the curved arrows tell you about electron movement to determine the product(s) of each reaction step.

Short Answer
Learning Materials
Features
Discover
Chapter 5: Q 10TP (page 190)
Use what the curved arrows tell you about electron movement to determine the product(s) of each reaction step.




All the tools & learning materials you need for study success - in one app.
Get started for free
Draw the skeletal structure of 3,3-dimethyl-7-(1-methyl ethyl)-6-(1-methyl propyl) decane.
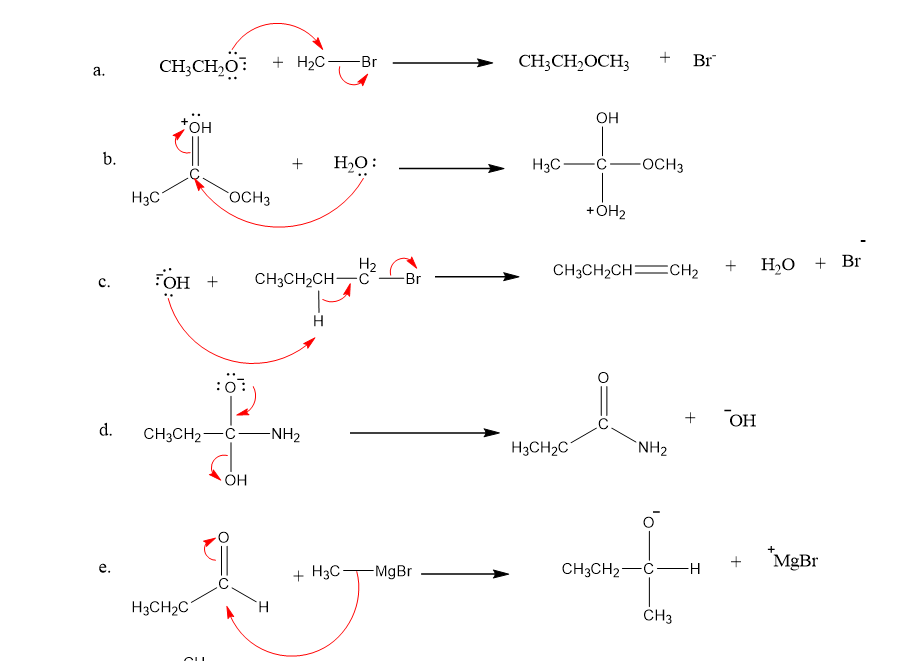
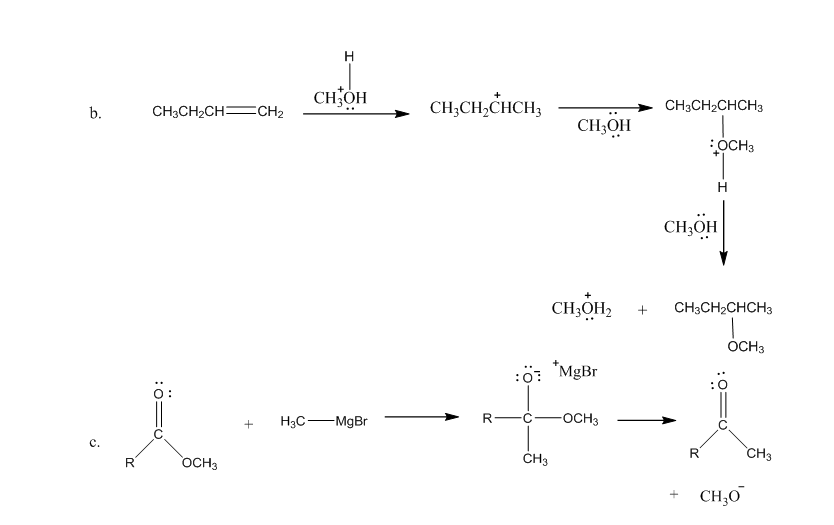
Draw curved arrows to show the movement of the electrons in each step of the following reaction sequences.
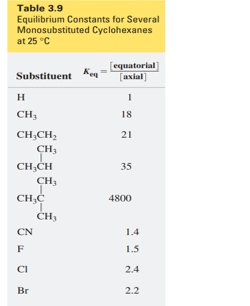
Question: a. Which of the monosubstituted cyclohexanes in Table 3.9 on p. 128 has a negative ∆G° for the conversion of an axial-substituted chair conformer to an equatorial-substituted chair conformer?
b.Which monosubstituted cyclohexane has the most negative ∆G° for this conversion?
c. Which monosubstituted cyclohexane has the greatest preference for an equatorial position?
d. Calculate ∆G° for the conversion of “axial” methylcyclohexane to “equatorial” methylcyclohexane at 25 °C.
Draw curved arrows to show the movement of the electrons that result in the formation of the given product(s). (Hint: Look at the structure of the product to see what bonds need to be formed and broken in order to arrive at the structure of the desired product.)
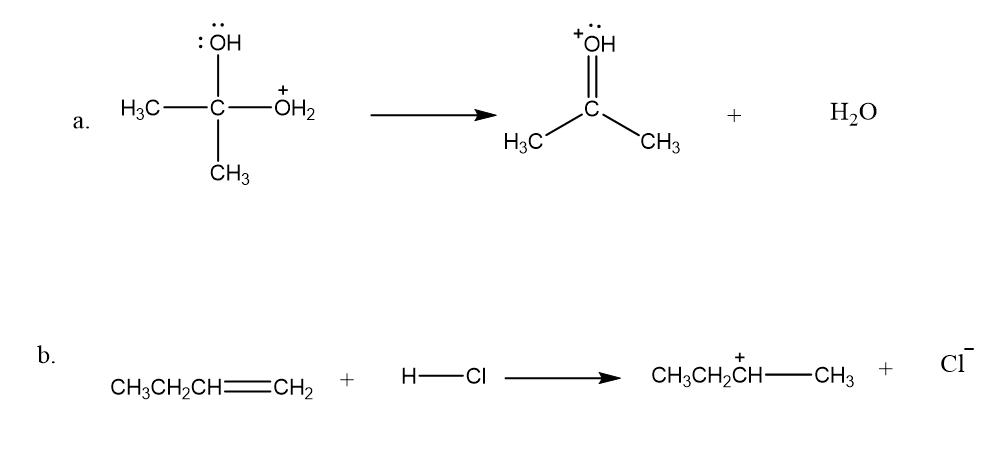
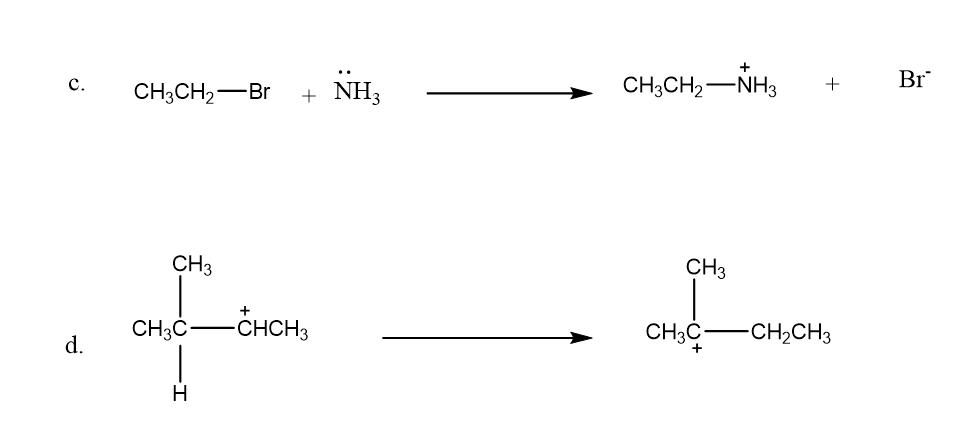
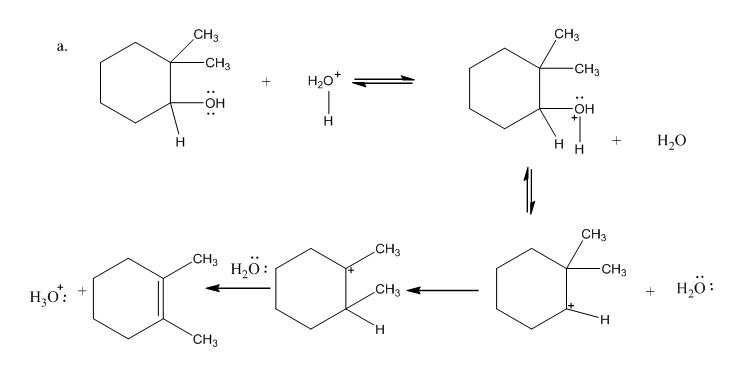
Draw curved arrows to show the movement of the electrons that result in formation of the given product(s).

What do you think about this solution?
We value your feedback to improve our textbook solutions.