Chapter 5: Q5-49P (page 222)
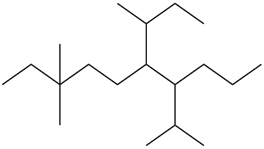
Draw the skeletal structure of 3,3-dimethyl-7-(1-methyl ethyl)-6-(1-methyl propyl) decane.
Short Answer
The skeletal structure is given as,
Learning Materials
Features
Discover
Chapter 5: Q5-49P (page 222)
Draw the skeletal structure of 3,3-dimethyl-7-(1-methyl ethyl)-6-(1-methyl propyl) decane.
The skeletal structure is given as,

All the tools & learning materials you need for study success - in one app.
Get started for free
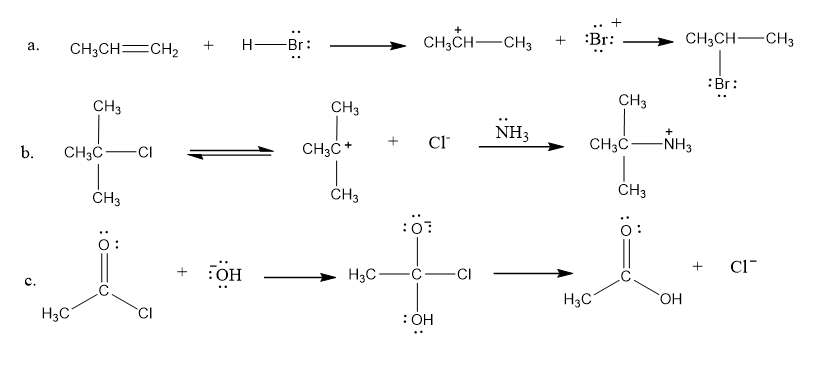
Draw curved arrows to show the movement of the electrons in each step of the following reaction sequences. (Hint: You can tell how to draw the arrows for each step by looking at the products that are formed in that step as a result of the movement of electrons.)
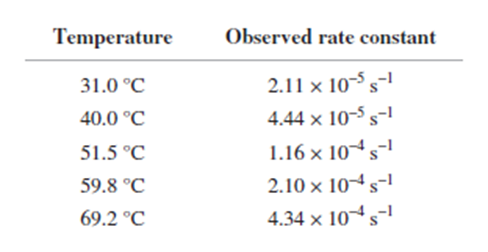
From the following rate constants, determined at five temperatures, calculate the experimental energy of activation and ΔG±,Δ��±,���Ի�Δ��±for the reaction at 30°C:
a. For a reaction with ∆H° = -12 kcal/mol and ∆S° = 0.01kcal mol-1 K-1, calculate the ∆G° and the equilibrium constant at: 1. 30 °C and 2. 150 °C.
b. How does ∆G° change as T increases?
c. How does Keq change as T increases?
Which bond is stronger? Briefly explain why.

In a reaction in which reactant A is in equilibrium with product B at
25 °C, what relative amounts of A and B are present at equilibrium if ΔG° at 25 °C is
a. 2.72 kcal/mol?
b. 0.65 kcal/mol?
c. -2.72 kcal/mol?
d. -0.65 kcal/mol?
What do you think about this solution?
We value your feedback to improve our textbook solutions.