Chapter 5: Q5-51P (page 190)
Which bond is stronger? Briefly explain why.

Short Answer
a. CH3-Cl bond is stronger.
b. I-Br bond is stronger.
Learning Materials
Features
Discover
Chapter 5: Q5-51P (page 190)
Which bond is stronger? Briefly explain why.

a. CH3-Cl bond is stronger.
b. I-Br bond is stronger.
All the tools & learning materials you need for study success - in one app.
Get started for free
Use what the curved arrows tell you about electron movement to determine the product(s) of each reaction step.
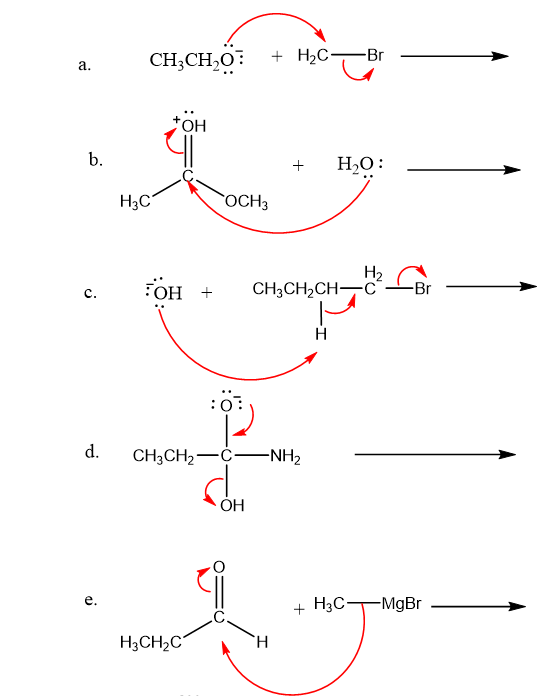
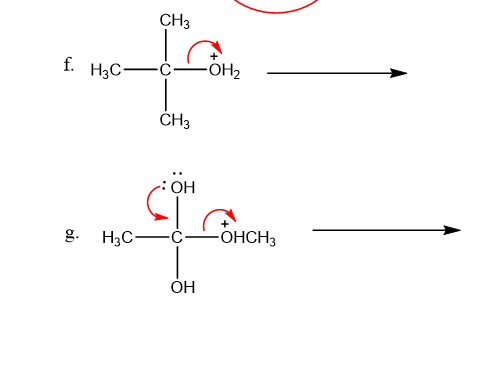
Draw curved arrows to show the movement of the electrons in each step of the following reaction sequences.

Draw structures for the following:
a. (2E,4E)-1-chloro-3-methyl-2,4-hexadiene
b. (3Z,5E)-4-methyl-3,5-nonadiene
c. (3Z,5Z)-4,5-dimethyl-3,5-nonadiene
d. (3E,5E)-2,5-dibromo-3,5-octadiene
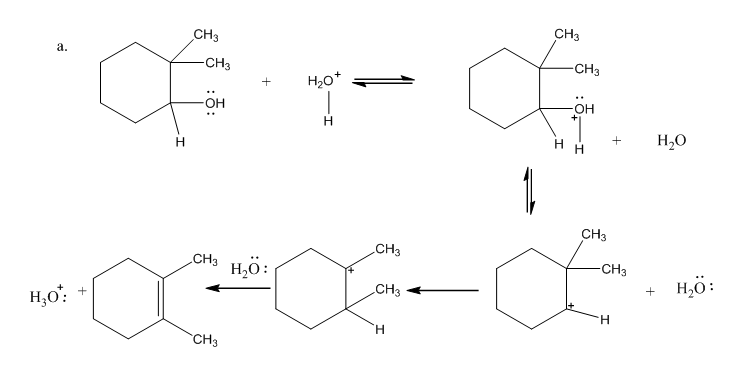
Draw curved arrows to show the movement of the electrons that result in formation of the given product(s).
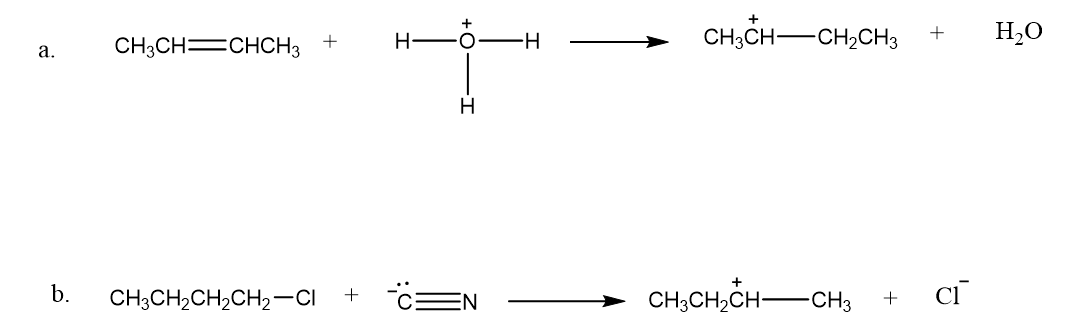

Draw curved arrows to show the flow of electrons responsible for the conversion of the reactants to products

What do you think about this solution?
We value your feedback to improve our textbook solutions.