Chapter 5: Q4TP (page 228)
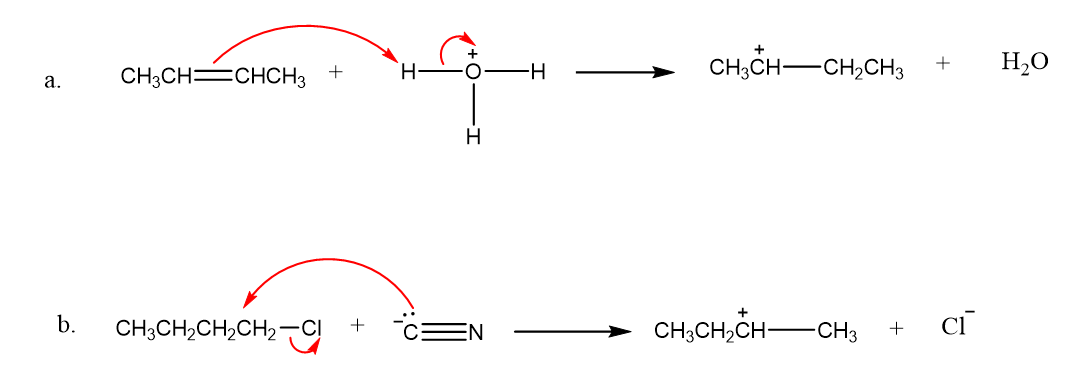
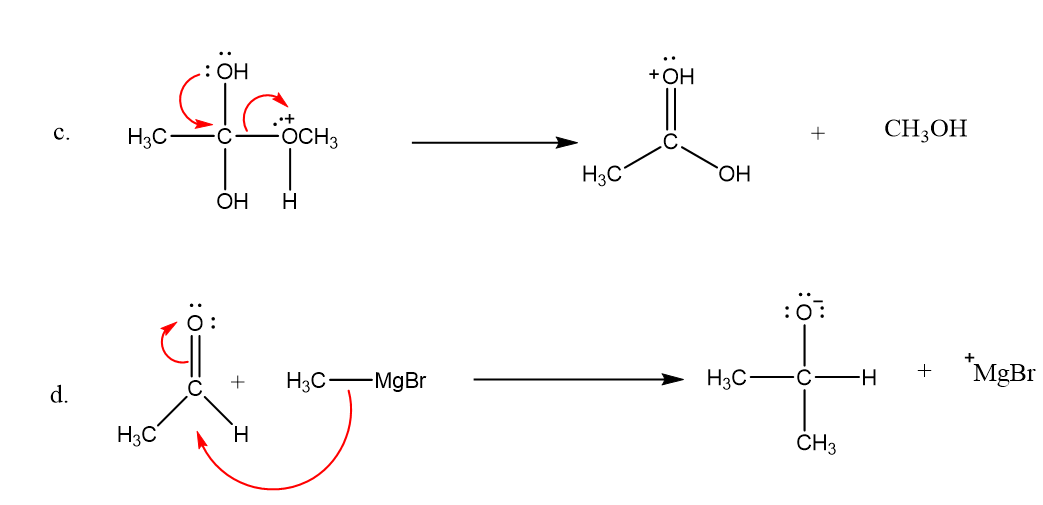
Draw curved arrows to show the movement of the electrons that result in formation of the given product(s).
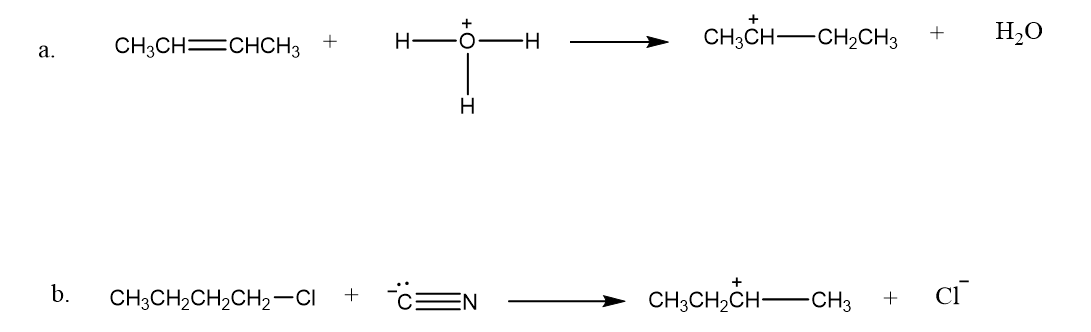
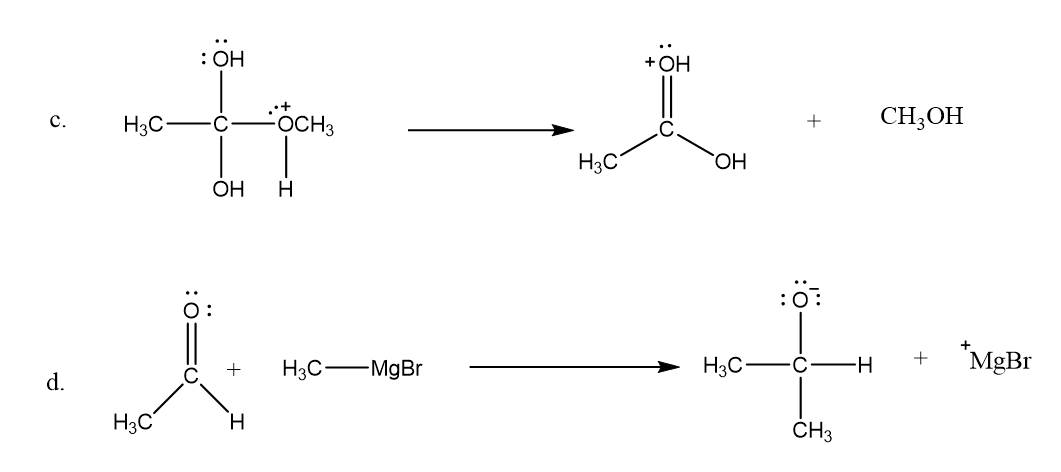
Short Answer
Learning Materials
Features
Discover
Chapter 5: Q4TP (page 228)
Draw curved arrows to show the movement of the electrons that result in formation of the given product(s).




All the tools & learning materials you need for study success - in one app.
Get started for free
a. Calculate the percentage of isopropylcyclohexane molecules that have the isopropyl substituent in an equatorial position at equilibrium. (Its ∆G° value at 25 °C is -2.1 kcal/mol.)
b. Why is the percentage of molecules with the substituent in an equatorial position greater for isopropylcyclohexane than for fluorocyclohexane?
Draw the condensed structure for each of the following:
a. (Z)-1,3,5-tribromo-2-pentene
b. (Z)-3-methyl-2-heptene
c. (E)-1,2-dibromo-3-isopropyl-2-hexene
d. vinyl bromide
e. 1,2-dimethylcyclopentene
f. diallyl amine
What is the molecular formula for each of the following?
For a reaction carried out at 25°C with an equilibrium constant of 1 × 10−3, in order to increase the equilibrium constant by a factor of 10:
a. How much must ΔG° change?
b. How much must ΔH° change if ΔS° = 0 kcal mol-1 K-1?
c. How much must ΔS° change if ΔH° = 0 kcal mol-1?
Question: Determine the degree of unsaturation and then draw possible structures for noncyclic compounds with the following molecular formulas:
a. C3H6
b. C3H4
c. C4H6
What do you think about this solution?
We value your feedback to improve our textbook solutions.