Chapter 5: Q9TP (page 230)
Draw curved arrows to show the movement of the electrons in each step of the following reaction sequences.
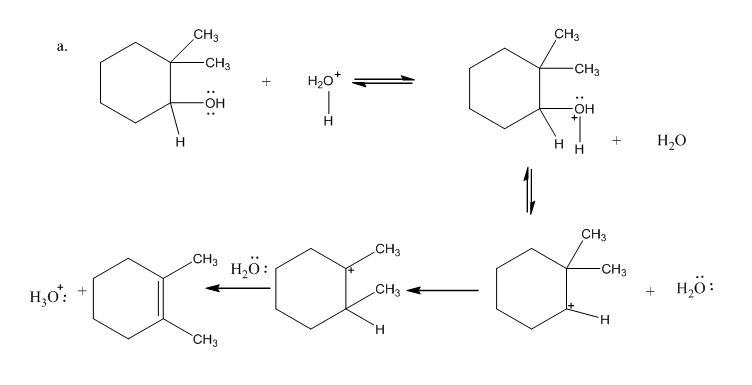
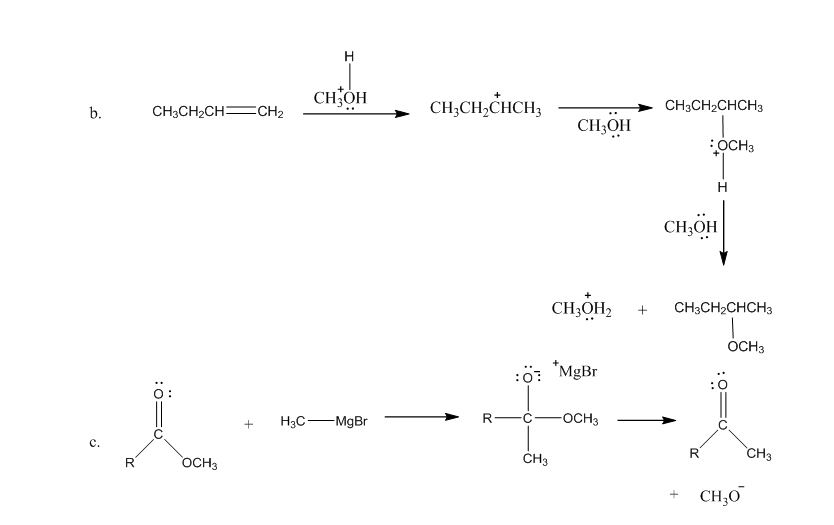
Short Answer
Learning Materials
Features
Discover
Chapter 5: Q9TP (page 230)
Draw curved arrows to show the movement of the electrons in each step of the following reaction sequences.




All the tools & learning materials you need for study success - in one app.
Get started for free
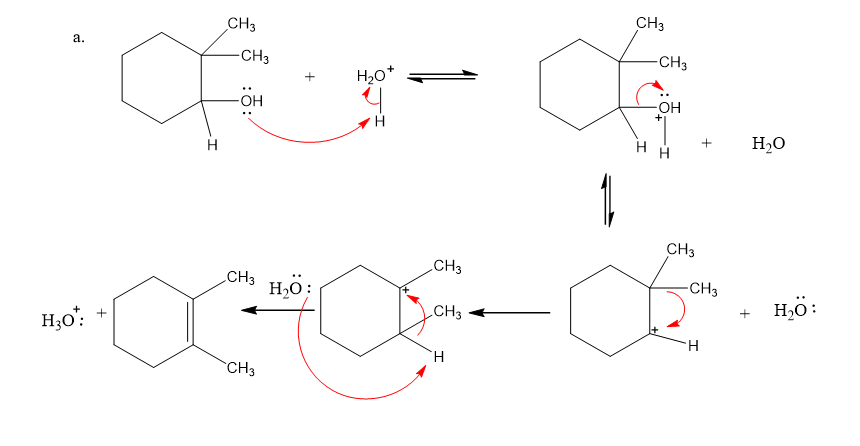
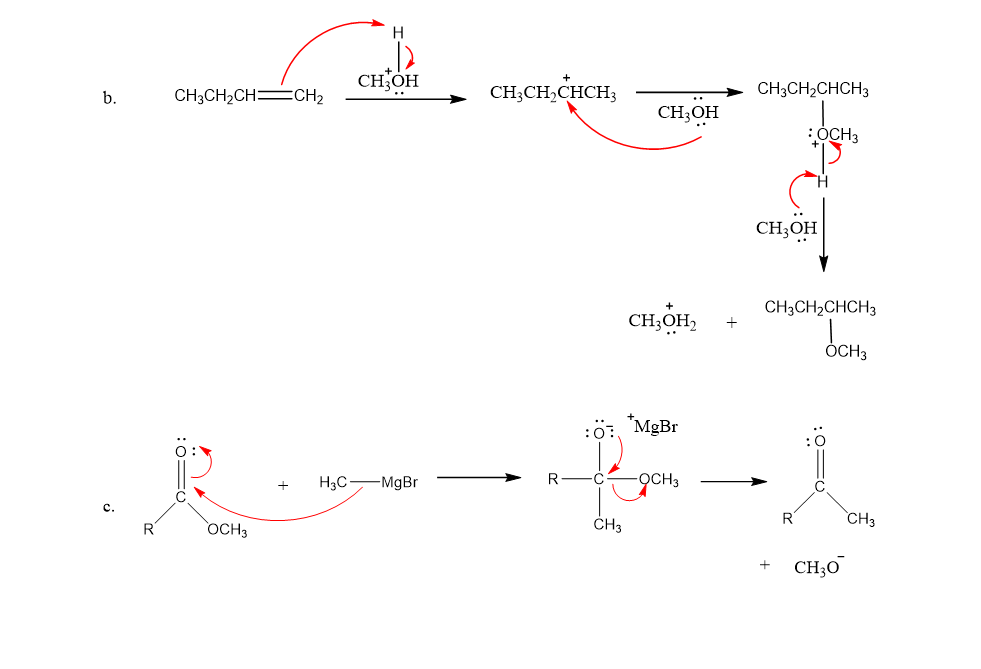
Draw curved arrows to show the flow of electrons responsible for the conversion of the reactants to products
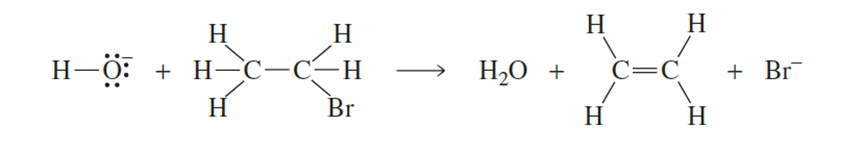
a. For a reaction with ∆H° = -12 kcal/mol and ∆S° = 0.01kcal mol-1 K-1, calculate the ∆G° and the equilibrium constant at: 1. 30 °C and 2. 150 °C.
b. How does ∆G° change as T increases?
c. How does Keq change as T increases?
Question: Draw the isomers for the following compounds and then name each one:
a. 2-methyl-2,4-hexadiene
b. 2,4-heptadiene
c. 1,3-pentadiene
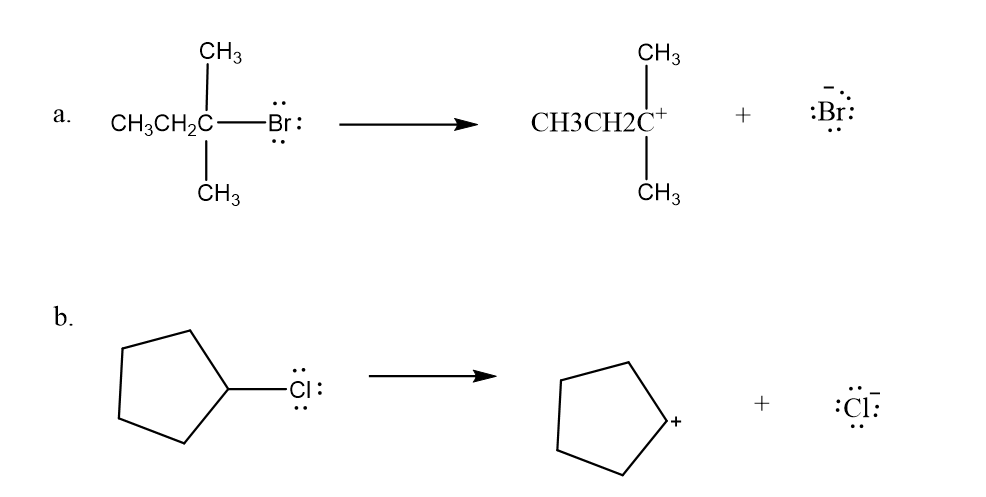
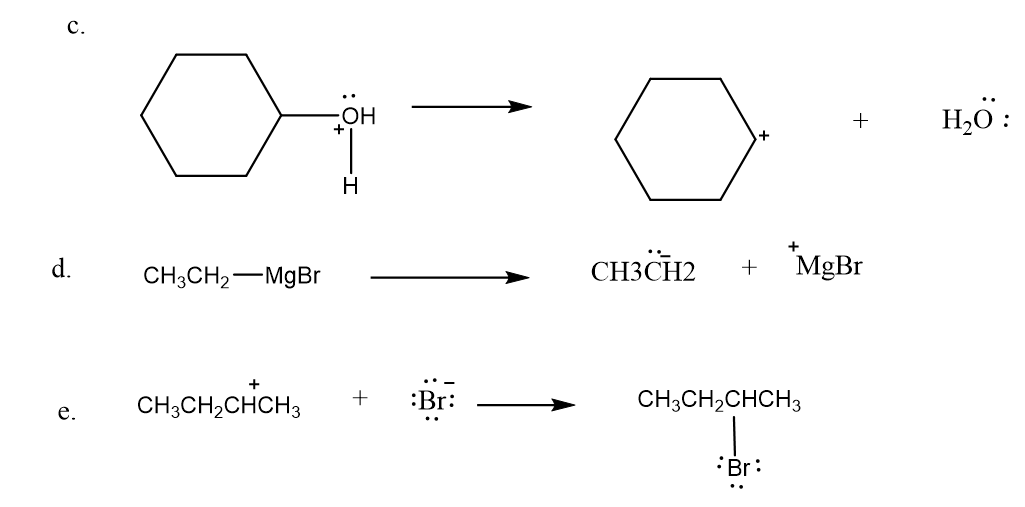
Draw curved arrows to show the movement of the electrons in the following reaction steps.
a. For which reaction in each set will ∆S° be more significant?
b. For which reaction will ∆S° be positive?
What do you think about this solution?
We value your feedback to improve our textbook solutions.