Chapter 5: Q21 P (page 205)
a. For which reaction in each set will ∆S° be more significant?
b. For which reaction will ∆S° be positive?
Short Answer
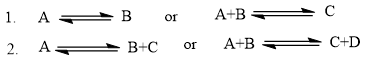
- Noun
Learning Materials
Features
Discover
Chapter 5: Q21 P (page 205)
a. For which reaction in each set will ∆S° be more significant?
b. For which reaction will ∆S° be positive?
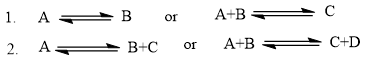
All the tools & learning materials you need for study success - in one app.
Get started for free
a. Draw the condensed structures and give the systematic names for all the alkenes with molecular formula C6H12, ignoring stereoisomers.
(Hint:There are 13.)
b. Which of the alkenes have Eand Zisomers?
c. Which of the alkenes is the most stable?
d. Which of the alkenes is the least stable?
Question: Determine the degree of unsaturation and then draw possible structures for noncyclic compounds with the following molecular formulas:
a. C3H6
b. C3H4
c. C4H6
Draw the structure of a hydrocarbon that has six carbon atoms and
a. three vinylic hydrogens and two allylic hydrogens.
b. three vinylic hydrogens and one allylic hydrogen.
c. three vinylic hydrogens and no allylic hydrogens.
Several studies have shown that b-carotene, a precursor of vitamin A, may play a role in preventing cancer. b-Carotene has a molecular formula of C40H56, and it contains two rings and no triple bonds. How many double bonds does it have?
Question: Determine the degree of unsaturation for hydrocarbons with the following molecular formulas:
a. C20H34 b. C8H16 c. C12H20 d. C40H56
What do you think about this solution?
We value your feedback to improve our textbook solutions.