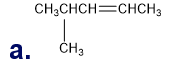
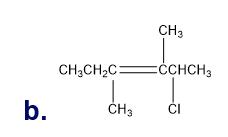
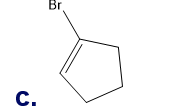
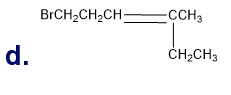
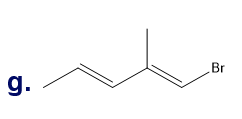
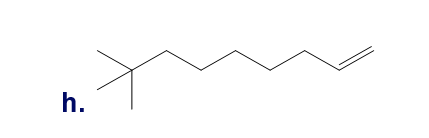
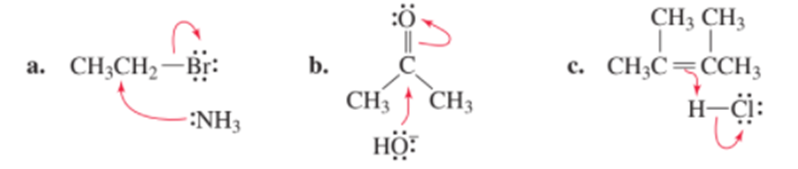
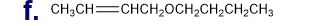Chapter 5: Q65P (page 224)
From the following rate constants, determined at five temperatures, calculate the experimental energy of activation and Δ��±,Δ��±,���Ի�Δ��±for the reaction at 30°C:
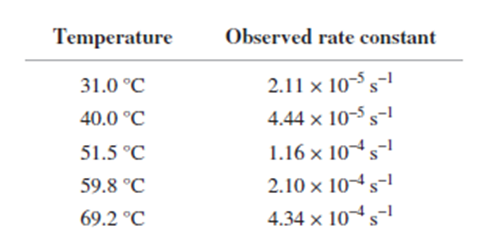
Short Answer
We have to findΔ��o, ΔHo, ΔSo
��o = 2.10 kcal/mol
ΔH° = 15.8 kcal/mol
ΔS° = 45 cal/(mol deg)