Chapter 5: Q14P (page 197)
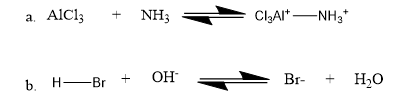
Question: Identify the nucleophile and the electrophile in the following acid–base reactions:
Short Answer
AlCl3is an electrophileand NH3 is a nucleophile.
The H+ of HBr is the electrophile and OH- is the nucleophile.
Learning Materials
Features
Discover
Chapter 5: Q14P (page 197)
Question: Identify the nucleophile and the electrophile in the following acid–base reactions:

AlCl3is an electrophileand NH3 is a nucleophile.
The H+ of HBr is the electrophile and OH- is the nucleophile.
All the tools & learning materials you need for study success - in one app.
Get started for free
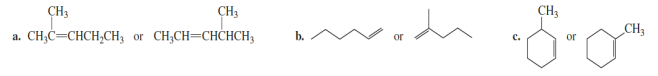
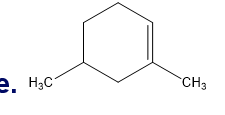
Which is more stable?
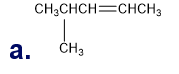
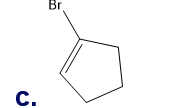
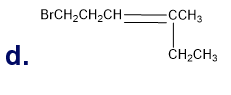
What is each compound’s systematic name?
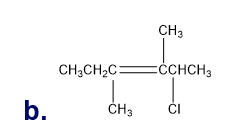
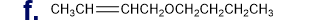

Question: Determine the degree of unsaturation for hydrocarbons with the following molecular formulas:
a. C20H34 b. C8H16 c. C12H20 d. C40H56
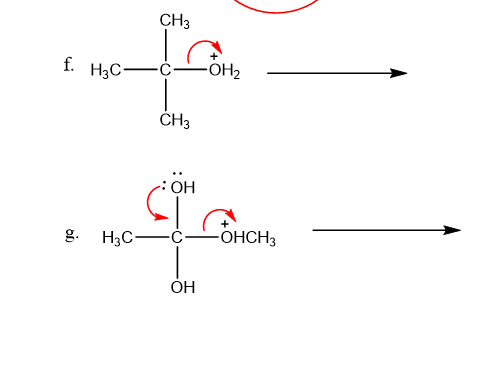
Question: Draw the products that would be obtained by following the incorrect arrows in the box entitled “A Few Words about Curved Arrows” and explain what is wrong with the structures you obtain
Use what the curved arrows tell you about electron movement to determine the product(s) of each reaction step.

What do you think about this solution?
We value your feedback to improve our textbook solutions.