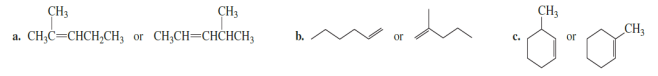
Chapter 5: Q40P (page 222)
Which is more stable?
Short Answer
a) 1st compound is more stable.
b) 2nd compound is more stable.
c) 2nd compound is more stable.
Learning Materials
Features
Discover
Chapter 5: Q40P (page 222)
Which is more stable?

a) 1st compound is more stable.
b) 2nd compound is more stable.
c) 2nd compound is more stable.
All the tools & learning materials you need for study success - in one app.
Get started for free
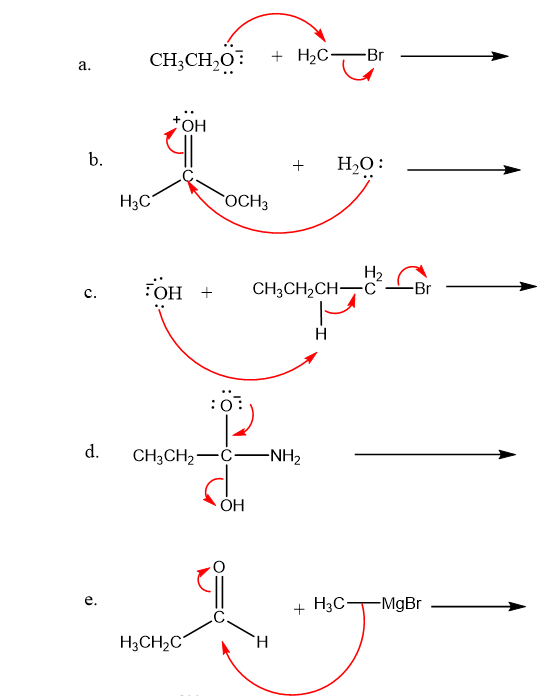
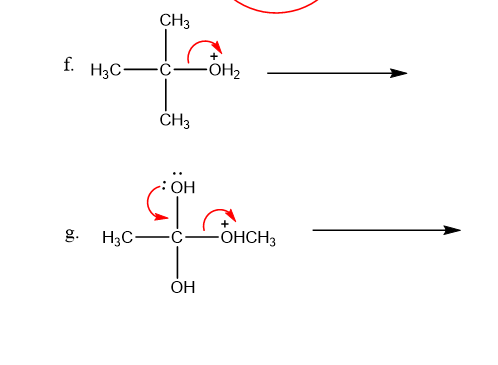
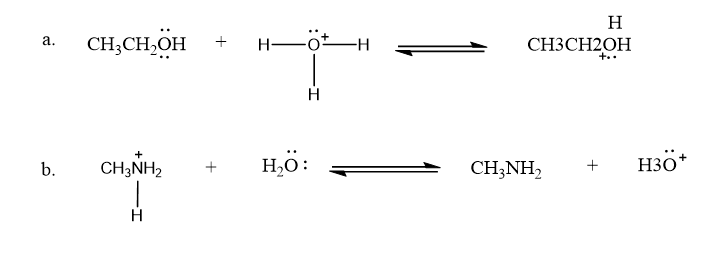
Use what the curved arrows tell you about electron movement to determine the product(s) of each reaction step.
a. For which reaction in each set will ∆S° be more significant?
b. For which reaction will ∆S° be positive?
Draw curved arrows to show the movement of the electrons that result in formation of the given product(s).

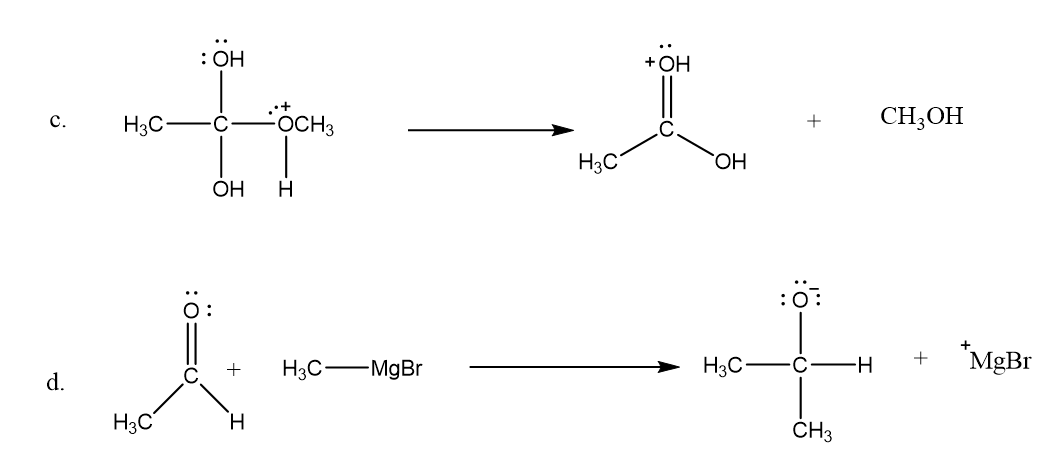
Draw curved arrows to show the movement of the electrons that result in formation of the given product(s).
For a reaction carried out at 25°C with an equilibrium constant of 1 × 10−3, in order to increase the equilibrium constant by a factor of 10:
a. How much must ΔG° change?
b. How much must ΔH° change if ΔS° = 0 kcal mol-1 K-1?
c. How much must ΔS° change if ΔH° = 0 kcal mol-1?
What do you think about this solution?
We value your feedback to improve our textbook solutions.