Chapter 23: Q8P (page 1076)
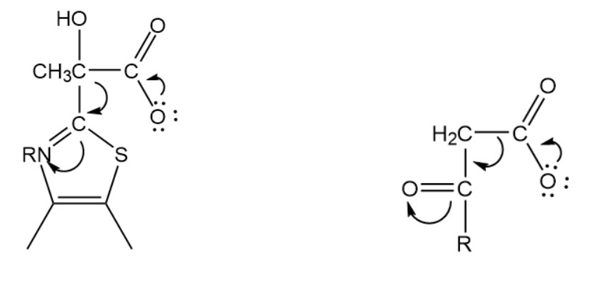
Draw structures that show the similarity between the decarboxylation of the pyruvate–TPP intermediate and the decarboxylation of a \(\beta \)-keto acid.
Short Answer
Learning Materials
Features
Discover
Chapter 23: Q8P (page 1076)
Draw structures that show the similarity between the decarboxylation of the pyruvate–TPP intermediate and the decarboxylation of a \(\beta \)-keto acid.

All the tools & learning materials you need for study success - in one app.
Get started for free
Explain why the hydrogens of the C-8 methyl group are more acidic than those of the C-7 methyl group.
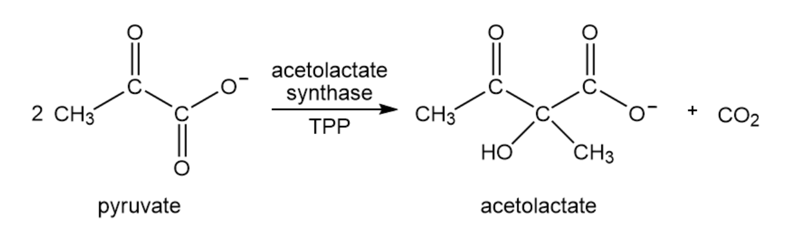
Acetolactate synthase is another TPP-requiring enzyme. It transfers the acyl group of pyruvate to another molecule of pyruvate, forming acetolactate. This is the first step in the biosynthesis of the amino acids valine and leucine. Propose a mechanism for this reaction.
Question: Do all triacylglycerols have the same number of asymmetric centers?
What are the products of the following reaction?
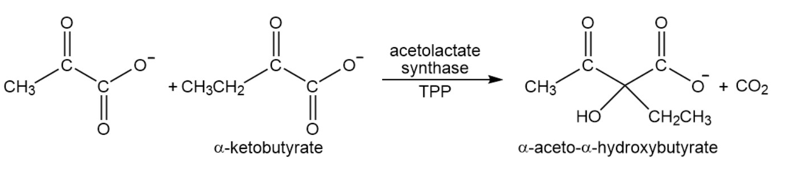
Acetolactatesynthasetransfers the acyl group of pyruvate to \(\alpha \)-ketobutyrate. This is the first step in the biosynthesis of the amino acid isoleucine. Propose a mechanism for this reaction.
What do you think about this solution?
We value your feedback to improve our textbook solutions.