Chapter 23: Q21P (page 1092)
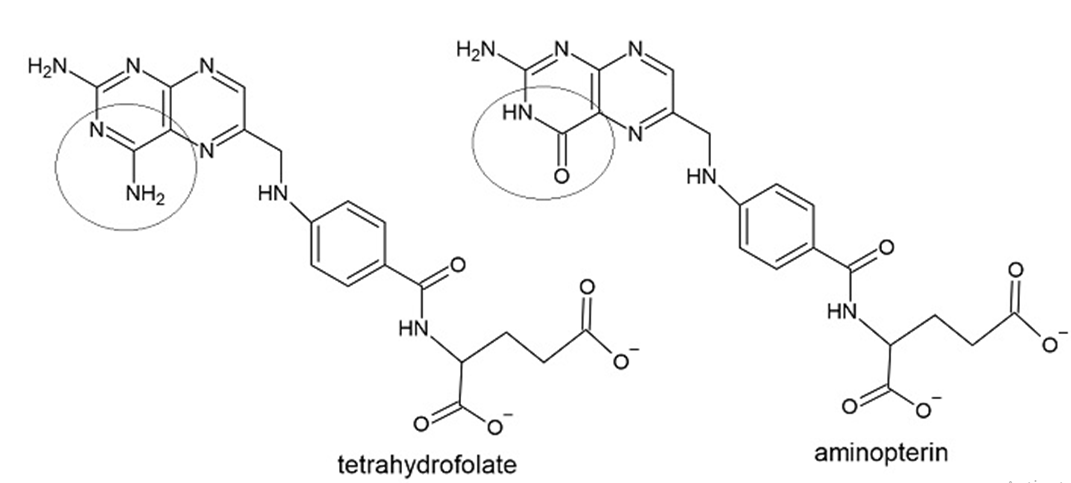
Question: How do the structures of tetrahydrofolate and aminopterin differ?
Short Answer
These structures differ by an amino group in tetrahydrofolate and keto group in aminopterin
Learning Materials
Features
Discover
Chapter 23: Q21P (page 1092)
Question: How do the structures of tetrahydrofolate and aminopterin differ?
These structures differ by an amino group in tetrahydrofolate and keto group in aminopterin

All the tools & learning materials you need for study success - in one app.
Get started for free
Question:Draw the products of the following reaction, where T is tritium:
The enzyme that catalyzes the bond cleavage reaction that converts serine to glycine removes the substituent (R) bonded to the \(\alpha \)-carbon in the first step of the reaction. Starting with PLP bound to serine in an imine linkage, propose a mechanism for this reaction. (Hint:The first step involves removal of the proton from serine’s OH group.)
Propose a mechanism for the -elimination reaction shown on page 1082.
Question: Propose a mechanism for the following reaction:
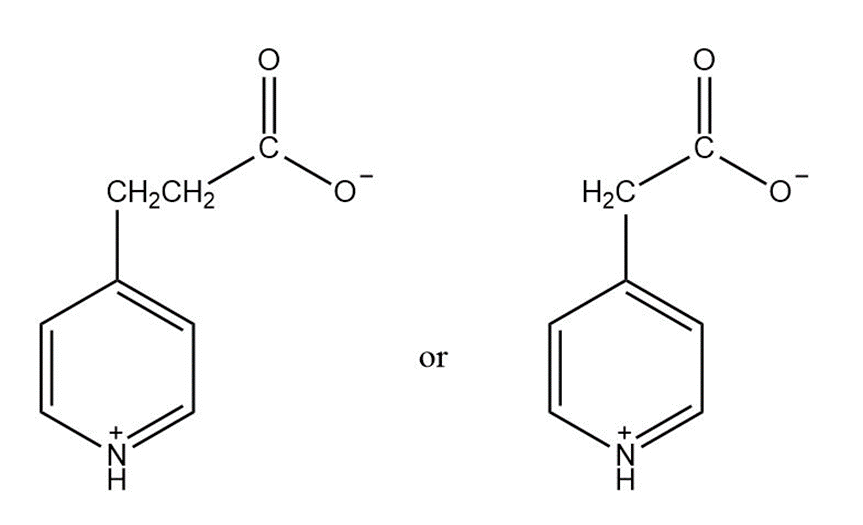
Which compound is more easily decarboxylated?
What do you think about this solution?
We value your feedback to improve our textbook solutions.