Chapter 1: Q-1-33P (page 2)
Predict the approximate bond angle in
- The methyl cation
- The methyl radical
- The methyl anion
Short Answer
- 1200
- 1200
- 1090
Learning Materials
Features
Discover
Chapter 1: Q-1-33P (page 2)
Predict the approximate bond angle in
All the tools & learning materials you need for study success - in one app.
Get started for free
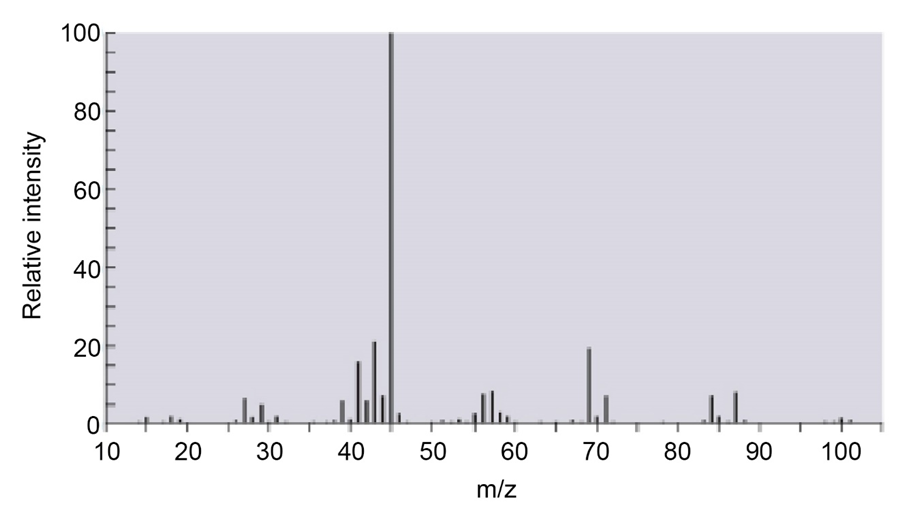
The mass spectrum for a compound with molecular weight of 102 is shown below. Its IR spectrum has a broad, strong absorption at 3600 cm–1 and a
medium absorption at 1360 cm–1.
a.Identify the compound.
b.Show the mechanism for formation of the peak at m/z= 84.
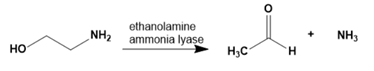
Question:Ethanolamine ammonia lyase, a coenzyme B12–requiring enzyme, catalyzes the following reaction. Propose a mechanism for this reaction
a. Draw two Lewis structures for C2H6O.
b. Draw three Lewis structures for C3H8O.
(Hint:The two Lewis structures in part a are constitutional isomers—molecules that have the same atoms but differ in the way the atoms are connected. The three Lewis structures in part b are also
constitutional isomers.)
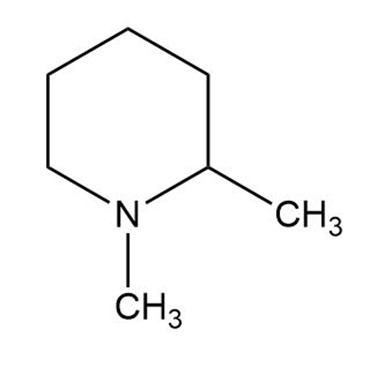
What products are obtained when the following tertiary amines react with hydrogen peroxide followed by heat?
a.
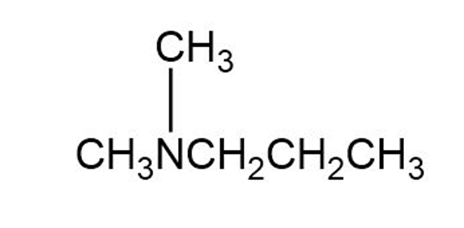
b.
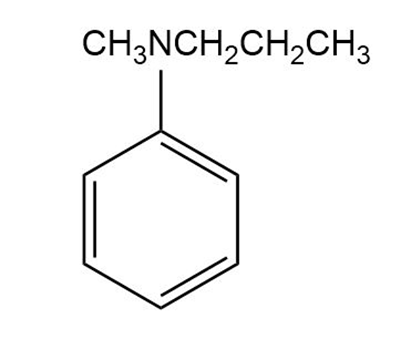
c.
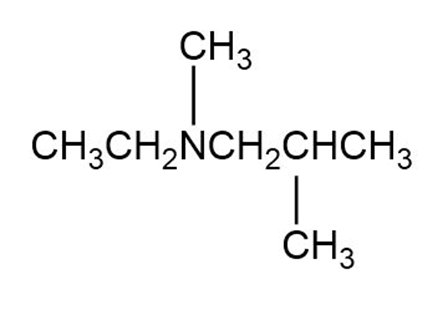
d.
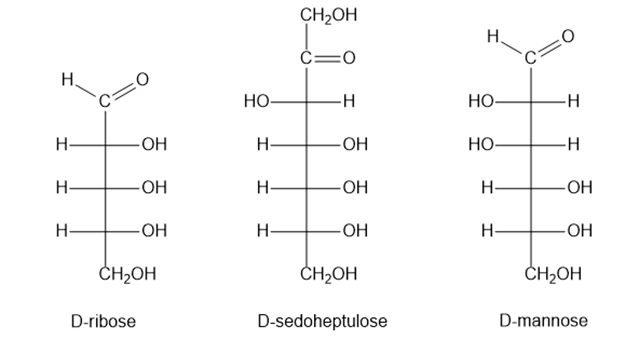
Classify the following monosaccharides:
What do you think about this solution?
We value your feedback to improve our textbook solutions.