Chapter 1: Q1P (page 2)
Classify the following monosaccharides:
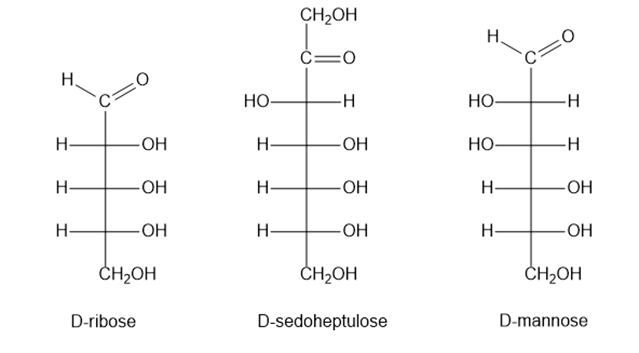
Short Answer
D-ribose is an aldopentose
D-sedoheptulose is a ketoheptose
D-mannose is an aldohexose
Learning Materials
Features
Discover
Chapter 1: Q1P (page 2)
Classify the following monosaccharides:

D-ribose is an aldopentose
D-sedoheptulose is a ketoheptose
D-mannose is an aldohexose
All the tools & learning materials you need for study success - in one app.
Get started for free
What is the hybridization of each of the carbon atoms in the following compound?
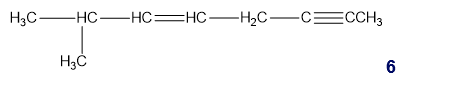
b. What is the hybridization of each of the C, N, and O atoms in Demerol and Prozac?(Generic names are not capitalized; trade names are capitalized.)
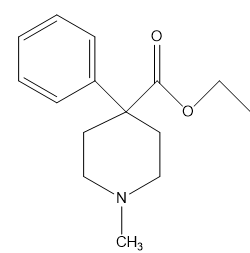
Demerol® : used to treat moderate to severe pain
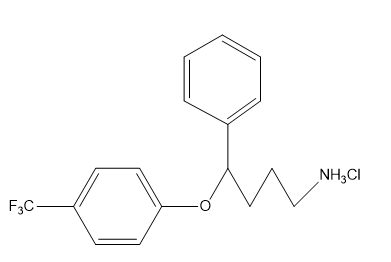
Prozac: used to treat depression, obsessive-compulsive disorder, some eating disorders, and panic attacks
Draw the structure for each of the following:
a. 2-bromo-4-iodophenol
b. 2,5-dinitrobenzaldehyde
c. 4-bromo-3-chloroaniline
Excess ammonia must be used when a primary amine is synthesized by reductive amination. What product will be obtained if the reaction is carried out with excess carbonyl compound?
Give each atom the appropriate formal charge:
Question:a. Draw a Lewis structure for each of the following:
1. CH3N2+ 2. HNO3 3. N3- 4. CH3CONH2 5. O3
b. Draw a structure for each of the species that shows approximate bond angles.
c. Which species have no dipole moment?
What do you think about this solution?
We value your feedback to improve our textbook solutions.