Chapter 1: Q-1-44P (page 2)
What is the hybridization of each of the carbon atoms in the following compound?
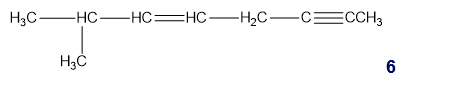
b. What is the hybridization of each of the C, N, and O atoms in Demerol and Prozac?(Generic names are not capitalized; trade names are capitalized.)
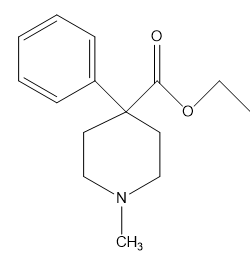
Demerol® : used to treat moderate to severe pain
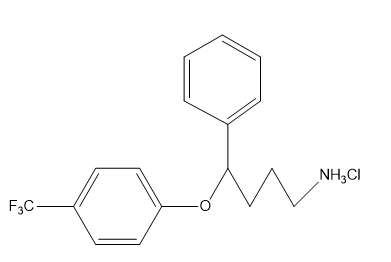
Prozac: used to treat depression, obsessive-compulsive disorder, some eating disorders, and panic attacks
Short Answer
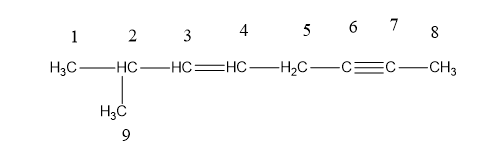
a.
C1= SP3
C2=SP3
C3=SP3
C4=SP3
C5=SP3
C6=SP3
C7=SP3
C8=SP3
C9=SP3
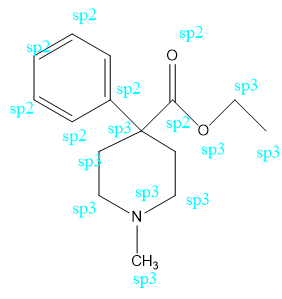
b. Demerol
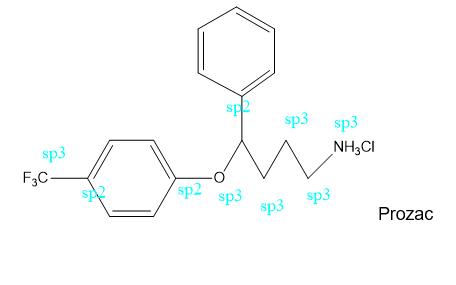


 HC=C-
HC=C- H2C=CH-
H2C=CH- H3C-CH2-
H3C-CH2-