Chapter 1: Q21P (page 2)
Rank the carbanions shown in the margin from most basic to least basic.
 HC=C-
HC=C-
 H2C=CH-
H2C=CH-
 H3C-CH2-
H3C-CH2-
Short Answer
H3C-CH2->H2C=CH->HC=C-
Learning Materials
Features
Discover
Chapter 1: Q21P (page 2)
Rank the carbanions shown in the margin from most basic to least basic.
 HC=C-
HC=C-
 H2C=CH-
H2C=CH-
 H3C-CH2-
H3C-CH2-
H3C-CH2->H2C=CH->HC=C-
All the tools & learning materials you need for study success - in one app.
Get started for free
Look at the relative position of each pair of atoms listed here in the periodic table. How many core electrons does each have? How many valence electrons does each have?
If the dipole moment of CH3F is 1.847 D and the dipole moment of CD3F is 1.858 D, which is more electronegative: hydrogen or deuterium?
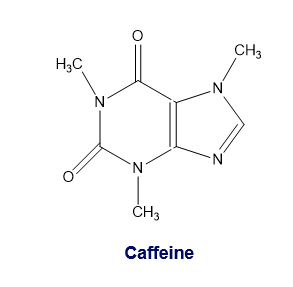
Caffeine is a natural insecticide found in the seeds and leaves of certain plants, where it kills insects that feed on the plant. Caffeine is extracted for human consumption from beans of the coffee plant, from Kola nuts, and from the leaves of tea plants. Because it stimulates the central nervous system, it temporarily prevents drowsiness. Add caffeine’s missing lone pairs to its structure.
Excess ammonia must be used when a primary amine is synthesized by reductive amination. What product will be obtained if the reaction is carried out with excess carbonyl compound?
a. Which bond will be longer?
b. Which bond will be stronger?
What do you think about this solution?
We value your feedback to improve our textbook solutions.