Chapter 1: Q-1-48P (page 2)
If the dipole moment of CH3F is 1.847 D and the dipole moment of CD3F is 1.858 D, which is more electronegative: hydrogen or deuterium?
Short Answer
Hydrogen is more electronegative than deuterium.
Learning Materials
Features
Discover
Chapter 1: Q-1-48P (page 2)
If the dipole moment of CH3F is 1.847 D and the dipole moment of CD3F is 1.858 D, which is more electronegative: hydrogen or deuterium?
Hydrogen is more electronegative than deuterium.
All the tools & learning materials you need for study success - in one app.
Get started for free
Excess ammonia must be used when a primary amine is synthesized by reductive amination. What product will be obtained if the reaction is carried out with excess carbonyl compound?
Imines can exist as stereoisomers. The isomers are named using the E, Z system of nomenclature (Section 4.2). The lone pair has the lowest priority.
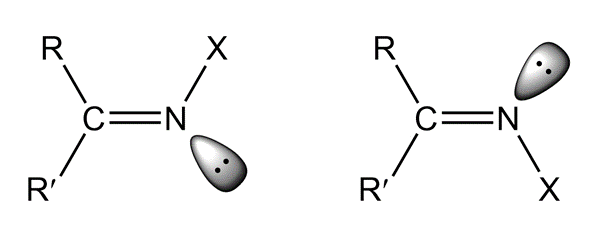
Draw the structure of each of the following compounds:
a. the (E)-hydrazone of benzaldehyde
b. the (Z)-oxime of propiophenone
The specific rotation of α -d-galactose is 150.7 and that of β -d-galactose is 52.8. When an aqueous mixture that was initially 70% α -d-galactose and 30% β -d-galactose reaches equilibrium, the specific rotation is 80.2. What is the percentage of α -d-galactose and β -d galactose at equilibrium?
After examining the potential maps for LiH, HF and H2, answer the following questions:
:(+)-Mandelic acid has a specific rotation of +158. What would be the observed specific rotation of each of the following mixtures?
a. 50% (-)-mandelic acid and 50% (+)-mandelic acid
b. 25% (-)-mandelic acid and 75% (+)-mandelic acid
c. 75% (-)-mandelic acid and 25% (+)-mandelic acid
What do you think about this solution?
We value your feedback to improve our textbook solutions.