Chapter 1: Q-1-49P (page 2)
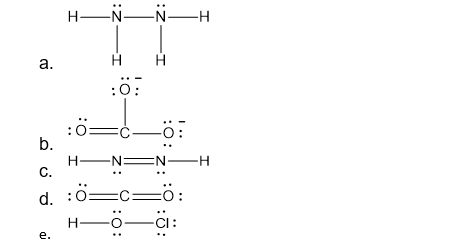
Draw a Lewis structure for each of the following:
- N2H4
- CO32-
- N2H2
- CO2
- HOCl
Short Answer
Learning Materials
Features
Discover
Chapter 1: Q-1-49P (page 2)
Draw a Lewis structure for each of the following:

All the tools & learning materials you need for study success - in one app.
Get started for free
Draw the ground-state electronic configuration for each of the following:
a. Mg
b.Ca2+
c. Ar
d. Mg2+
Look at the relative position of each pair of atoms listed here in the periodic table. How many core electrons does each have? How many valence electrons does each have?
An unknown disaccharide gives a positive Tollens’ test. A glycosidase hydrolyzes it to d-galactose and d-mannose. When the disaccharide is treated with methyl iodide and Ag2O and then hydrolyzed with dilute HCl, the products are 2,3,4,6-tetra-O-methylgalactose and 2,3,4-tri-O-methylmannose. Propose
a structure for the disaccharide.
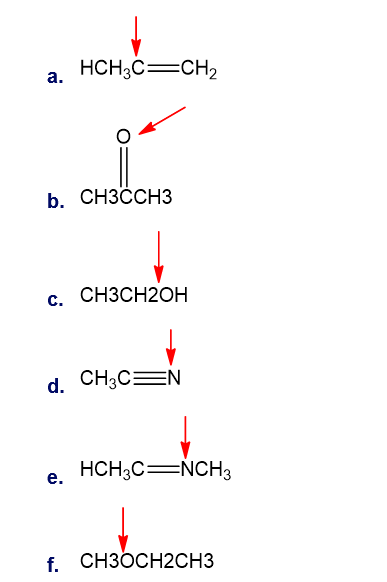
Question: What is the hybridisation of indicated atoms in each of the following?
Compare the electrostatic potential maps for methane, ammonia and water. Which is the most polar molecule? Which is the least polar?

What do you think about this solution?
We value your feedback to improve our textbook solutions.