Chapter 1: Q54P (page 2)
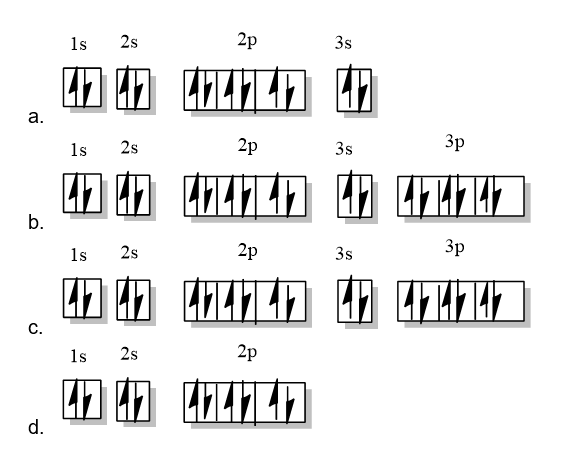
Draw the ground-state electronic configuration for each of the following:
a. Mg
b.Ca2+
c. Ar
d. Mg2+
Short Answer
Learning Materials
Features
Discover
Chapter 1: Q54P (page 2)
Draw the ground-state electronic configuration for each of the following:
a. Mg
b.Ca2+
c. Ar
d. Mg2+

All the tools & learning materials you need for study success - in one app.
Get started for free
Indicate the kind of molecular orbital (σ ,σ *,π ,π *) that results when the two atomic orbitals are combined:
Describe the orbital used in bonding and the bond angles in the following compounds:
a. CH3O-
b. CO2
c. H2CO
d. N2
e. BF3
Put a number in each of the blanks.
:(+)-Mandelic acid has a specific rotation of +158. What would be the observed specific rotation of each of the following mixtures?
a. 50% (-)-mandelic acid and 50% (+)-mandelic acid
b. 25% (-)-mandelic acid and 75% (+)-mandelic acid
c. 75% (-)-mandelic acid and 25% (+)-mandelic acid
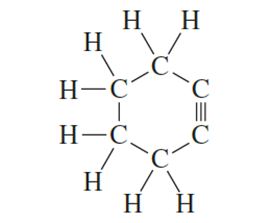
Explain why the following compound is not stable
What do you think about this solution?
We value your feedback to improve our textbook solutions.