Chapter 1: Q75P (page 49)
Explain why the following compound is not stable
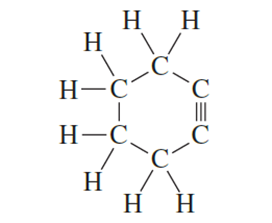
Short Answer
The compound is not stable due to angle strain in its conformer along the triple bond. There is about 60° angle strain.
Learning Materials
Features
Discover
Chapter 1: Q75P (page 49)
Explain why the following compound is not stable

The compound is not stable due to angle strain in its conformer along the triple bond. There is about 60° angle strain.
All the tools & learning materials you need for study success - in one app.
Get started for free
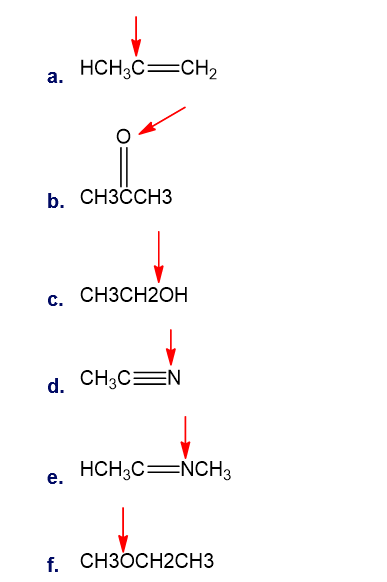
Question: What is the hybridisation of indicated atoms in each of the following?
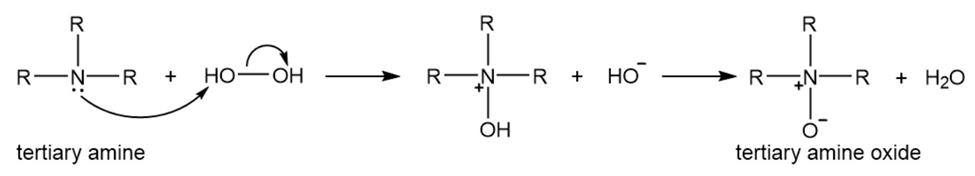
A tertiary amine reacts with hydrogen peroxide to form a tertiary amine oxide.
Tertiary amine oxides undergo a reaction similar to the Hofmann elimination reaction (Section 10.10), called a Cope elimination. In this reaction, a tertiary amine oxide, rather than a quaternary ammonium ion, undergoes elimination. A strong base is not needed for a Cope elimination because the amine oxide acts as its own base.
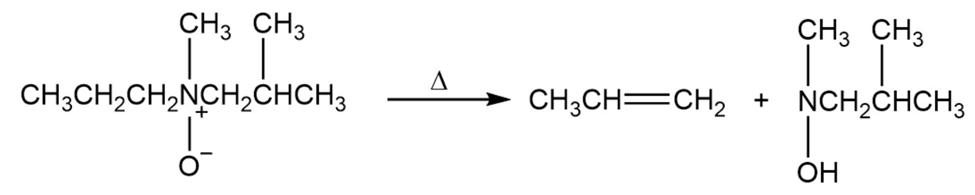
Does the Cope elimination have an alkene-like transition state or a carbanion-like transition state?
Use the symbol and to show the direction of polarity of the bond between carbon and oxygen.
:(+)-Mandelic acid has a specific rotation of +158. What would be the observed specific rotation of each of the following mixtures?
a. 50% (-)-mandelic acid and 50% (+)-mandelic acid
b. 25% (-)-mandelic acid and 75% (+)-mandelic acid
c. 75% (-)-mandelic acid and 25% (+)-mandelic acid
Rank the bonds from the most polar to least polar.
a. C - O,C - F,C - N
b. C - Cl > C - Br > C - I
c. H - O,H - N,H - C
d. C - H,C - C,C - N
What do you think about this solution?
We value your feedback to improve our textbook solutions.