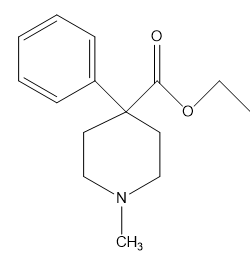
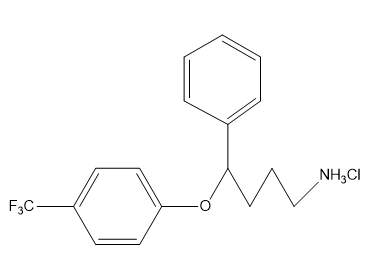
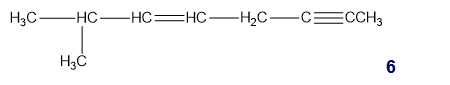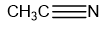Chapter 1: Q31P (page 2)
A tertiary amine reacts with hydrogen peroxide to form a tertiary amine oxide.
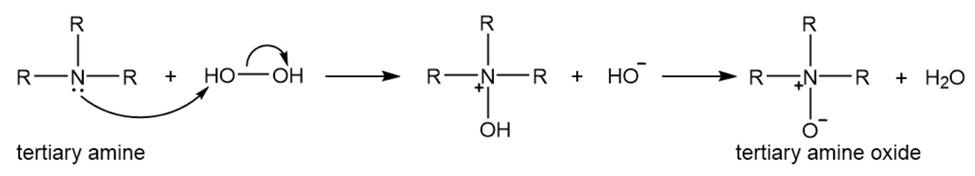
Tertiary amine oxides undergo a reaction similar to the Hofmann elimination reaction (Section 10.10), called a Cope elimination. In this reaction, a tertiary amine oxide, rather than a quaternary ammonium ion, undergoes elimination. A strong base is not needed for a Cope elimination because the amine oxide acts as its own base.
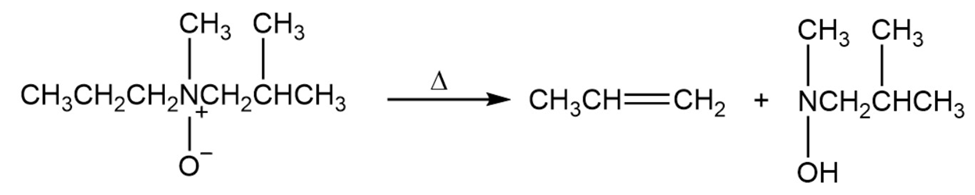
Does the Cope elimination have an alkene-like transition state or a carbanion-like transition state?
Short Answer
.
Cope elimination transition state is an alkene-like transition state.