Chapter 1: Q26P (page 25)
Indicate the kind of molecular orbital (σ ,σ *,π ,π *) that results when the two atomic orbitals are combined:
Short Answer
��.π *
��.π
��.σ
��.σ *
Learning Materials
Features
Discover
Chapter 1: Q26P (page 25)
Indicate the kind of molecular orbital (σ ,σ *,π ,π *) that results when the two atomic orbitals are combined:
��.π *
��.π
��.σ
��.σ *
All the tools & learning materials you need for study success - in one app.
Get started for free
Explain why the following compound is not stable
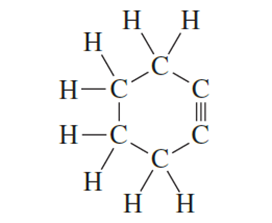
Explain why σ bond formed by overlap of s-orbital with SP3orbital of carbon is stronger than abond formed by the overlap of an s orbital with a p orbital of carbon.
Calculate the percentages of α-d-glucose and β-d-glucose present at equilibrium from the specific rotations of α-d-glucose, β-d-glucose, and the equilibrium mixture. Compare your values with those given in Section 20.10. (Hint:The specific rotation of the mixture equals the specific rotation of α-d-glucose times the fraction of glucose present in the α-form plus the specific rotation of β-d-glucose times the fraction of glucose present in the β-form.)
Draw condensed structures for the compounds represented by the following models
(black = C, grey = H, red = O, blue = N, and green = Cl):
Question: What is the major product of each of the following reactions?
a.
b.
c.
d.
What do you think about this solution?
We value your feedback to improve our textbook solutions.