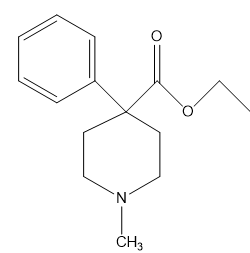
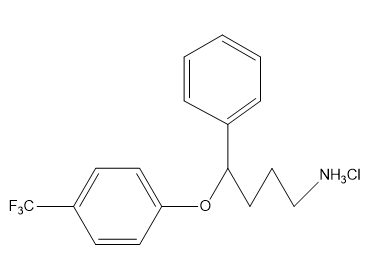
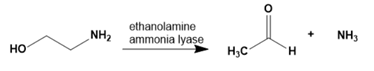
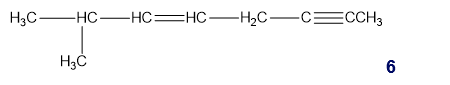Chapter 1: Q65P (page 2)
Calculate the percentages of α-d-glucose and β-d-glucose present at equilibrium from the specific rotations of α-d-glucose, β-d-glucose, and the equilibrium mixture. Compare your values with those given in Section 20.10. (Hint:The specific rotation of the mixture equals the specific rotation of α-d-glucose times the fraction of glucose present in the α-form plus the specific rotation of β-d-glucose times the fraction of glucose present in the β-form.)
Short Answer
The calculation indicates that 36% is in the α-form and 64% is in the β-form. This agrees with the values in Section 21.10.