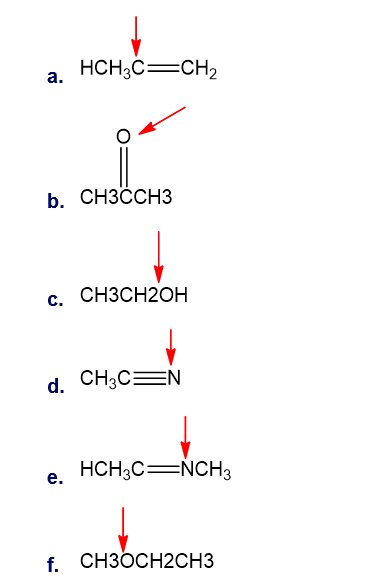
Chapter 1: Q13P (page 2)
Explain why HCI has a smaller dipole moment than HF,even though the H -CI bond is longer than the H-F bond.
Short Answer
HF has a larger electronegativity difference compared to that of HCI and thus it has a smaller dipole moment despite its longer bond length.