Chapter 1: Q-1-41P (page 2)
Which of the bonds in a carbon-oxygen double bond has more effective orbital overlap: the bond σ or the bond π ?
Short Answer
The σ bond in the carbon-oxygen double bond has a more effective orbital overlap.
Learning Materials
Features
Discover
Chapter 1: Q-1-41P (page 2)
Which of the bonds in a carbon-oxygen double bond has more effective orbital overlap: the bond σ or the bond π ?
The σ bond in the carbon-oxygen double bond has a more effective orbital overlap.
All the tools & learning materials you need for study success - in one app.
Get started for free
A ketone can be prepared from the reaction of a nitrile with a Grignard reagent. Describe the intermediate formed in this reaction, and show how it can be converted to a ketone.
Predict the approximate bond angles for
Draw the following orbitals:
a. 3sorbital
b. 4sorbitalc.
c. 3porbital
a.Which of the species have bond angles of 109.5°?
b. Which of the species have bond angles of 120°?
H2O H3O+ +CH3 BF3 NH3 +NH4 -CH3
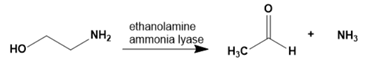
Question:Ethanolamine ammonia lyase, a coenzyme B12–requiring enzyme, catalyzes the following reaction. Propose a mechanism for this reaction
What do you think about this solution?
We value your feedback to improve our textbook solutions.