Chapter 1: Q24P (page 21)
Draw the following orbitals:
a. 3sorbital
b. 4sorbitalc.
c. 3porbital
Learning Materials
Features
Discover
Chapter 1: Q24P (page 21)
Draw the following orbitals:
a. 3sorbital
b. 4sorbitalc.
c. 3porbital
All the tools & learning materials you need for study success - in one app.
Get started for free
a.Which of the species have bond angles of 109.5°?
b. Which of the species have bond angles of 120°?
H2O H3O+ +CH3 BF3 NH3 +NH4 -CH3
Rank the carbanions shown in the margin from most basic to least basic.
 HC=C-
HC=C-
 H2C=CH-
H2C=CH-
 H3C-CH2-
H3C-CH2-
Question:a. Draw a Lewis structure for each of the following:
1. CH3N2+ 2. HNO3 3. N3- 4. CH3CONH2 5. O3
b. Draw a structure for each of the species that shows approximate bond angles.
c. Which species have no dipole moment?
Explain why HCI has a smaller dipole moment than HF,even though the H -CI bond is longer than the H-F bond.
The following compound has only one asymmetric centre. Why then does it have four stereoisomers?
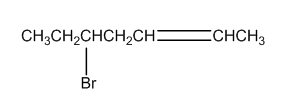
What do you think about this solution?
We value your feedback to improve our textbook solutions.