Chapter 1: Q27P (page 28)
What orbitals are used to form 10 sigma bonds in propane( CH 3CH 2CH3)?
Short Answer
In propane 2, sigma bonds are formed by SP3 -SP3overlap and rest eight sigma bonds are formed by the overlap of SP3with s-orbital.
Learning Materials
Features
Discover
Chapter 1: Q27P (page 28)
What orbitals are used to form 10 sigma bonds in propane( CH 3CH 2CH3)?
In propane 2, sigma bonds are formed by SP3 -SP3overlap and rest eight sigma bonds are formed by the overlap of SP3with s-orbital.
All the tools & learning materials you need for study success - in one app.
Get started for free
A ketone can be prepared from the reaction of a nitrile with a Grignard reagent. Describe the intermediate formed in this reaction, and show how it can be converted to a ketone.
Draw the condensed structure of a compound that contains only carbon and hydrogen atoms and that has
a. Three sp3 hybridized carbons.
b. One sp3hybridized carbon and two sp2 hybridized carbons.
c. Two sp3 hybridized carbons and two sp hybridized carbons.
Convert the models in problem 20 to skeletal structures.
Question:Are the following pairs identical, enantiomers, diastereomers, or constitutional isomers?
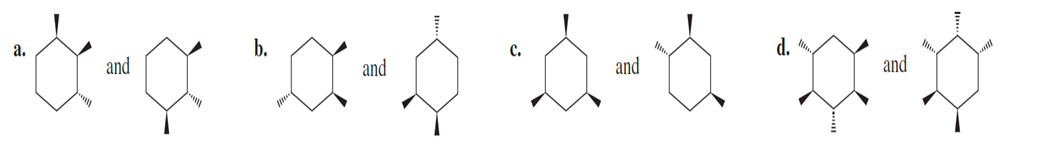
Which of the following molecules would you expect to have a dipole moment of zero? To answer parts g and h, you may need to review the Problem Solving Strategy on p. 39.

What do you think about this solution?
We value your feedback to improve our textbook solutions.