Chapter 1: Q-92P (page 2)
Question:Are the following pairs identical, enantiomers, diastereomers, or constitutional isomers?
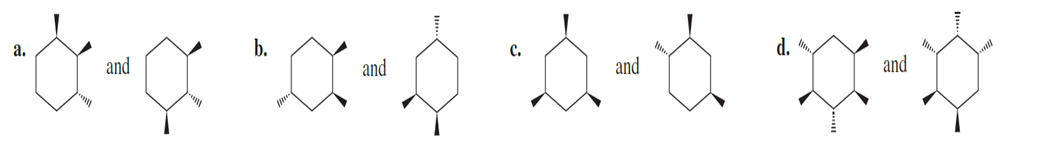
Short Answer
- They are diastereomers,
- They are identical
- They are Constitutional isomers.
- They are diastereomers,
Learning Materials
Features
Discover
Chapter 1: Q-92P (page 2)
Question:Are the following pairs identical, enantiomers, diastereomers, or constitutional isomers?

All the tools & learning materials you need for study success - in one app.
Get started for free
Indicate the kind of molecular orbital (σ ,σ *,π ,π *) that results when the two atomic orbitals are combined:
For each of the given species:a. Draw its Lewis structure.b. Describe the orbitals used by each carbon atom in bonding and indicate the approximate bond angles.
1.H2CO2
2. HCN
3. CCl4
4. H2CO3
Explain why protonating aniline dramatically affects the compound’s UV spectrum, whereas protonating pyridine has only a small effect on that compound’s UV spectrum.
Determine the partial negative charge on the fluorine atom in a C-F bond. The bond length is 1.39 1.39A* , and the bond dipole moment is 1.06D. The charge on an electron is 4.80⨯10-10esu.
Question: What is the major product of each of the following reactions?
a.
b.
c.
d.
What do you think about this solution?
We value your feedback to improve our textbook solutions.