Chapter 1: Q9P (page 2)
Which of the following has
- The most polar bond?
- The least polar bond?
- NaI, LiBr, Cl2, KCI
Short Answer
- KCI is most polar.
- CI2is least polar.
Learning Materials
Features
Discover
Chapter 1: Q9P (page 2)
Which of the following has
All the tools & learning materials you need for study success - in one app.
Get started for free
Draw the following orbitals:
a. 3sorbital
b. 4sorbitalc.
c. 3porbital
Question:a. Draw a Lewis structure for each of the following:
1. CH3N2+ 2. HNO3 3. N3- 4. CH3CONH2 5. O3
b. Draw a structure for each of the species that shows approximate bond angles.
c. Which species have no dipole moment?
What orbitals are used to form 10 sigma bonds in propane( CH 3CH 2CH3)?
After examining the potential maps for LiH, HF and H2, answer the following questions:
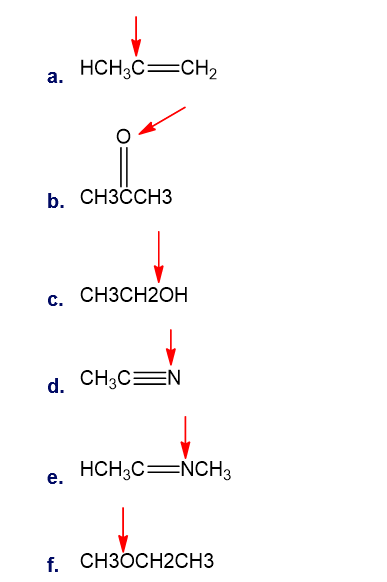
Question: What is the hybridisation of indicated atoms in each of the following?
What do you think about this solution?
We value your feedback to improve our textbook solutions.