Chapter 1: Q-1-46P (page 2)
Which of the following molecules would you expect to have a dipole moment of zero? To answer parts g and h, you may need to review the Problem Solving Strategy on p. 39.

Short Answer
BeCl2, BF3, have zero dipole moment.
Learning Materials
Features
Discover
Chapter 1: Q-1-46P (page 2)
Which of the following molecules would you expect to have a dipole moment of zero? To answer parts g and h, you may need to review the Problem Solving Strategy on p. 39.

BeCl2, BF3, have zero dipole moment.
All the tools & learning materials you need for study success - in one app.
Get started for free
There are three constitutional isomers with molecular formula C2H2Cl2. Draw their structures. Which one does not have a dipole moment?
Draw the lone-pair electrons that are not shown in the following condensed structures:
a. CH3CH2NH2
b. CH3NHCH3
c. CH3CH2OH
d. CH3OCH3
e. CH3CH2Cl
f. HONH2
Question: What is the major product of each of the following reactions?
a.
b.
c.
d.
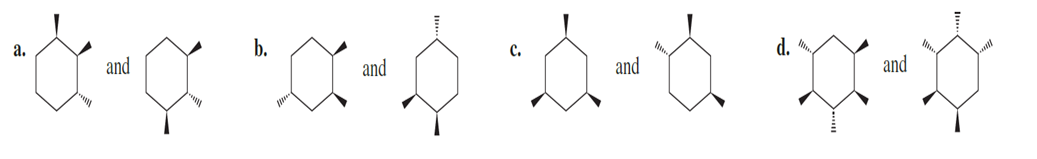
Question:Are the following pairs identical, enantiomers, diastereomers, or constitutional isomers?
Excess ammonia must be used when a primary amine is synthesized by reductive amination. What product will be obtained if the reaction is carried out with excess carbonyl compound?
What do you think about this solution?
We value your feedback to improve our textbook solutions.