Chapter 1: Q55P (page 2)
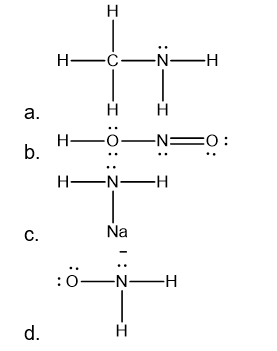
Draw a Lewis structure for each of the following:
a. CH3NH2
b. HNO3
c. NaNH2
d. NH2O-
Short Answer
Learning Materials
Features
Discover
Chapter 1: Q55P (page 2)
Draw a Lewis structure for each of the following:
a. CH3NH2
b. HNO3
c. NaNH2
d. NH2O-

All the tools & learning materials you need for study success - in one app.
Get started for free
Question:PLP can catalyze both \(\alpha ,\beta - \)-elimination reactions (Problem 33) and \(\beta ,\gamma - \)-elimination reactions. Propose a mechanism for the following PLP-catalyzed
\(\beta ,\gamma - \) elimination reaction:
Change the following condensed structure to Kekulẻ structures:
a. CH3NH(CH2)2CH3
b.(CH3)2CHCl
c.(CH3)3CBr
d.(CH3)3C(CH2)3CHO
Convert the models in problem 20 to skeletal structures.
Rank the carbanions shown in the margin from most basic to least basic.
 HC=C-
HC=C-
 H2C=CH-
H2C=CH-
 H3C-CH2-
H3C-CH2-
Draw the condensed structure of a compound that contains only carbon and hydrogen atoms and that has
a. Three sp3 hybridized carbons.
b. One sp3hybridized carbon and two sp2 hybridized carbons.
c. Two sp3 hybridized carbons and two sp hybridized carbons.
What do you think about this solution?
We value your feedback to improve our textbook solutions.