Chapter 1: Q22P (page 19)
Change the following condensed structure to Kekulẻ structures:
a. CH3NH(CH2)2CH3
b.(CH3)2CHCl
c.(CH3)3CBr
d.(CH3)3C(CH2)3CHO
Learning Materials
Features
Discover
Chapter 1: Q22P (page 19)
Change the following condensed structure to Kekulẻ structures:
a. CH3NH(CH2)2CH3
b.(CH3)2CHCl
c.(CH3)3CBr
d.(CH3)3C(CH2)3CHO
All the tools & learning materials you need for study success - in one app.
Get started for free
Draw a Lewis structure for each of the following:
a. CH3NH2
b. HNO3
c. NaNH2
d. NH2O-
What is the hybridisation of each of the carbon and oxygen atoms in vitamin c?
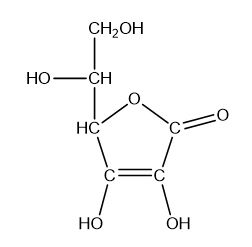
Draw Lewis structure of the following
a.CH3CHO
b.CH3OCH3
c.CH3COOH
Question:a. Draw a Lewis structure for each of the following:
1. CH3N2+ 2. HNO3 3. N3- 4. CH3CONH2 5. O3
b. Draw a structure for each of the species that shows approximate bond angles.
c. Which species have no dipole moment?
Predict the approximate bond angles:
a. The C-N-H bond angle in (CH3)2NH2+
b. The C-O-H bond angle in CH3OH .
c. The C-N-H bond angle in (CH3)NH
d. The C-N-H bond angle in (CH3)2NH
What do you think about this solution?
We value your feedback to improve our textbook solutions.