Chapter 1: Q58P (page 2)
Draw Lewis structure of the following
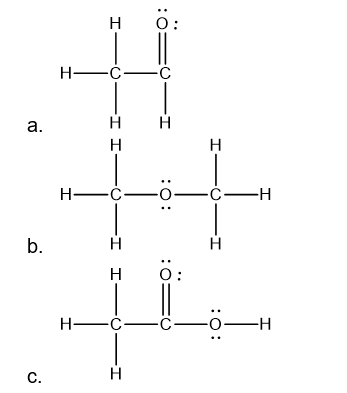
a.CH3CHO
b.CH3OCH3
c.CH3COOH
Short Answer
Answer:
Learning Materials
Features
Discover
Chapter 1: Q58P (page 2)
Draw Lewis structure of the following
a.CH3CHO
b.CH3OCH3
c.CH3COOH
Answer:

All the tools & learning materials you need for study success - in one app.
Get started for free
The compounds commonly known as “amino acids” are actually -aminocarboxylic acids (Section 21.0). What carbonyl compounds should be used to synthesize the two amino acids shown here?
Compare the electrostatic potential maps for methane, ammonia and water. Which is the most polar molecule? Which is the least polar?

An unknown disaccharide gives a positive Tollens’ test. A glycosidase hydrolyzes it to d-galactose and d-mannose. When the disaccharide is treated with methyl iodide and Ag2O and then hydrolyzed with dilute HCl, the products are 2,3,4,6-tetra-O-methylgalactose and 2,3,4-tri-O-methylmannose. Propose
a structure for the disaccharide.
Give each atom the appropriate formal charge:
Question:a. Draw a Lewis structure for each of the following:
1. CH3N2+ 2. HNO3 3. N3- 4. CH3CONH2 5. O3
b. Draw a structure for each of the species that shows approximate bond angles.
c. Which species have no dipole moment?
What do you think about this solution?
We value your feedback to improve our textbook solutions.