Chapter 1: Q16P (page 2)
Give each atom the appropriate formal charge:
Short Answer
We have to give each atom appropriate formal charge:
Learning Materials
Features
Discover
Chapter 1: Q16P (page 2)
Give each atom the appropriate formal charge:
We have to give each atom appropriate formal charge:
All the tools & learning materials you need for study success - in one app.
Get started for free
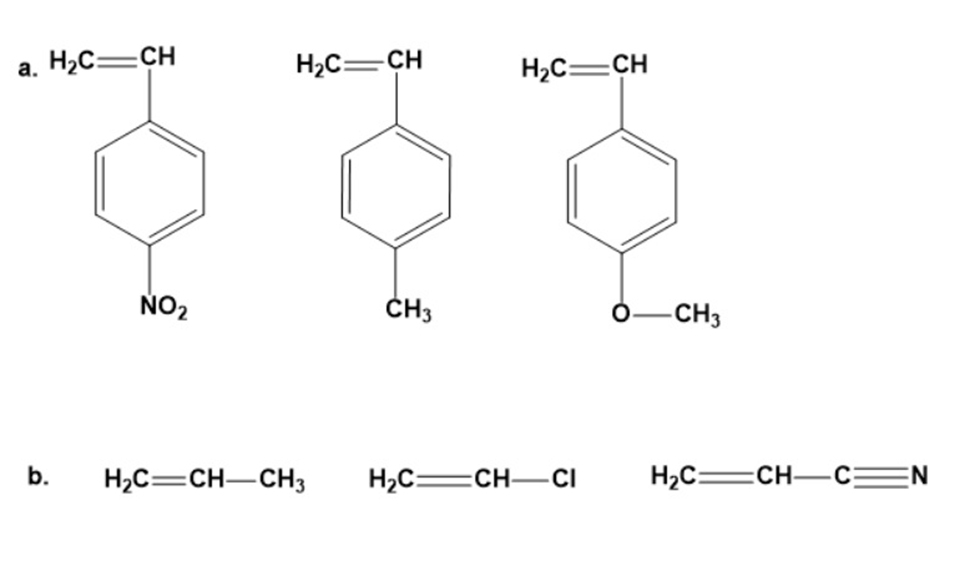
Rank the following groups of monomers from most able to least able to undergo anionic polymerization:
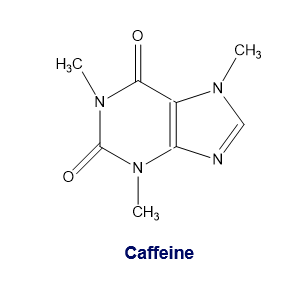
Caffeine is a natural insecticide found in the seeds and leaves of certain plants, where it kills insects that feed on the plant. Caffeine is extracted for human consumption from beans of the coffee plant, from Kola nuts, and from the leaves of tea plants. Because it stimulates the central nervous system, it temporarily prevents drowsiness. Add caffeine’s missing lone pairs to its structure.
Draw the following orbitals:
a. 3sorbital
b. 4sorbitalc.
c. 3porbital
Rank the carbanions shown in the margin from most basic to least basic.
 HC=C-
HC=C-
 H2C=CH-
H2C=CH-
 H3C-CH2-
H3C-CH2-
a. Find potassium (K) in the periodic table and predict how many valence electrons it has.b. What orbital does the unpaired electron occupy?
What do you think about this solution?
We value your feedback to improve our textbook solutions.